48uep6bbphidvals|451
48uep6bbph|2000F98CTab_Articles|Fulltext
Splenic rupture is a rare life threatening complication usually
occurring after trauma. Atraumatic splenic rupture (ASR) can
occur in diseased (pathologic rupture) or normal spleen
(spontaneous rupture).[1,2,3] ASR is associated with varied
etiologies including, neoplastic, infectious, inflammatory, noninfectious,
drug-and treatment-related and mechanical
disorders.[1,2,3] A recent study showed secondary metastatic
neoplastic disorders and pancreatitis to be associated in 3.8%
and 9.2% of ASR respectively.[2]
We report here two cases of ASR. The first case describes
ASR subsequent to disseminated duodenal adenocarcinoma,
while the second is a rare case of acute on chronic pancreatitis.
To the best of our knowledge the former is the first case of its
kind to be reported in literature.
Case 1
A 68 year old male presented with history of severe abdominal
pain in left hypochondrium and epigastrium. There was no
history of trauma. He had tachycardia, pallor and mild tenderness in left hypochondrium. His hemoglobin was 6.5
gm/dl and serum alkaline phosphatase (SAP) level raised by
1.5 times. Contrast enhanced computed tomography (CECT)
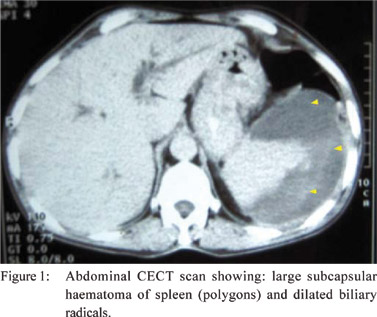
scan (Figure 1) showed a large subcapsular splenic hematoma
and dilatation of biliary radicals.
Esophagogastroduodenoscopy revealed an irregular ulcer of
less then 1cm size in second part of duodenum (D2).
Histopathological examination (H&E) of biopsy from the ulcer
showed chronic inflammatory cells without any evidence of
malignancy. The patient was instituted intravenous fluids,
analgesics, proton pump inhibitor, antibiotics and blood
transfusion. The patient showed clinical improvement and
reduction in the size of the hematoma and was discharged on
treatment for H. pylori infection and analgesics.
Six weeks later, he presented with vomiting suggestive of
gastric outlet obstruction (GOO). He also had mild pain in his
right flank and right hip. His hemoglobin was 8.5 gm/dl and
SAP level raised by 3 times. Esophagogastroduodenoscopy
revealed polypoidal lesion with friability in the D2 segment.
Histopathology of the duodenal lesion showed moderately
differentiated duodenal adenocarcinoma. CECT scan with
angiography showed infiltrative growth involving second and
third part of duodenum, circumferential thickening of gastric
wall, dilatations of common bile duct (CBD), biliary radicals
and main pancreatic duct (MPD), perinephric and retroperitoneal collection encasing the right renal hilum,
destructive lesion in right femoral head and small resolving
splenic haematoma. A diagnosis of disseminated duodenal
adenocarcinoma with femoral head metastasis and resolving
splenic hematoma was made. Treatment options including
palliative surgery, chemotherapy and enteral stenting were not opted because of his poor general condition and refusal to
give consent. Patient was managed with feeding jejunostomy,
analgesics and nutritional supplements. Five weeks later, the
patient died because of general debility.
Case 2
A 45 year old chronic alcoholic male presented with six days
history of moderate epigastric pain radiating to the back and
two days history of sudden, severe left hypochondrial pain
radiating to the left shoulder. There was no history of trauma.
His two-month old medical records revealed hemoglobin of
11gm/dl and ultrasonographic evidence of chronic pancreatitis.
He had tachycardia, pallor and tender left hypochondrium and
epigastrium. His hemoglobin was 4.5 gm/dl and serum amylase
raised by 2.5 times. CECT scan showed a 8 × 10 × 10 cm3
heterogeneous lesion along the splenic capsule compressing
the spleen and the posterior wall of stomach, mild atrophic
pancreas with dilated MPD and presence of fluid along the
pancreatic tail, ascites and left sided pleural effusion. Peritoneal
fluid aspiration revealed hemoperitoneum. He required
laparotomy which revealed a hematoma extending from splenic
parenchyma through the capsule into the perisplenic area
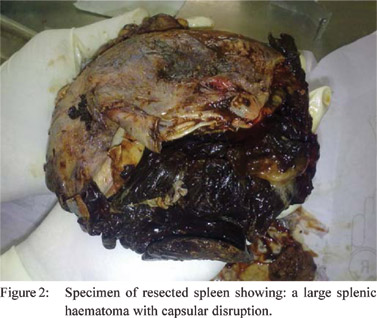
(Figure 2) and presence of free fluid along the edematous
pancreatic tail. Splenectomy was performed with satisfactory postoperative course. Pathological study showed capsular
disruption with adherent blood clots, subcapsular hemorrhage
and a tear in the spleen tissue with hemorrhages. He is currently
on alcohol deaddiction therapy, pancreatic enzyme supplements
and oral analgesics and is under our follow up since last four
months.
Discussion
ASR associated with neoplasms are mostly caused by
hematological malignancies however, malignancies of various
solid organs like ovary, lungs, skin, stomach, liver, pancreas,
urinary bladder, prostate, rectum and breast have been rarely
described.[1,2,3] There is no case report of ASR associated with
duodenal adenocarcinoma. The pathological processes
described for ASR in hematological malignancies include
capsular distention due to infiltration of the splenic
parenchyma and capsule, coagulation defects and splenic
infarct with subsequent capsular hemorrhage.[1,4,5] However,
the mechanisms implicated in solid tumors are direct invasion
or metastases and disruption of the splenic blood flow
secondary to thrombosis.[1,6]
Duodenal adenocarcinoma is a rare gastrointestinal
malignancy, having tendency of local invasion and metastasis
commonly to lymph nodes, liver, peritoneum and lung. At
presentation, over 45% of patients may have metastasis.[7]
The systemic spread of tumor cells into the spleen is the
possible explanation of rupture. Metastasis to bone is unusual
and the locations of bone metastases are not well described.[8,9]
We did not find any report of metastasis to femoral head.ASR
is a rare complication of pancreatitis.[10,11,12,13] The exact etiology
and natural history of this complication is not well described.
The likely mechanisms of splenic rupture in pancreatitis include
splenic vessels thrombosis, dissection of a pseudocyst into
the splenic hilum, splenic artery pseudoaneurysm erosion by
the pseudocyst contents, extension of the inflammatory
process from the pancreatic tail into the splenic hilum and
perisplenitis.[11] Treatment strategies for ASR include total
splenectomy, organ-preserving surgery or conservative
measures.[2] ASR carries a mortality rate of 12.2%, but
splenomegaly, advancing age and neoplastic disorders are
associated with higher mortality.[2]
References
- Debnath D, Valerio D. Atraumatic rupture of the spleen in adults.
J R Coll Surg Edinb. 2002;47:437–45.
- Renzulli P, Hostettler A, Schoepfer AM, Gloor B, Candinas D.
Systematic review of atraumatic splenic rupture. Br J Surg.
2009;96:1114–21.
- Görg C, Cölle J, Görg K, Prinz H, Zugmaier G. Spontaneous
rupture of the spleen: ultrasound patterns, diagnosis and followup.
Br J Radiol. 2003;76:704–11.
- Canady MR, Welling RE, Strobel SL. Splenic rupture in leukemia.
J Surg Oncol. 1989;41:194–7.
- Giagoundis AA, Burk M, Meckenstock G, Koch AJ, Schneider
W. Pathologic rupture of the spleen in hematologic malignancies:
two additional cases. Ann Haematol. 1996;73:297–302.
- Smith WM, Lucas JG, Frankel WL. Splenic rupture: a rare
presentation of pancreatic carcinoma. Arch Pathol Lab
Med. 2004;128:1146–50.
- Adedeji OA, Trescoli-Serrano C, Garcia-Zarco M. Primary
duodenal carcinoma. Postgrad Med J. 1995;71:354–8.
- Ghormley RK, Valls JE. Metastasis to bone from carcinoma of
the gastrointestinal tract. J Bone Joint Surg. 1939;21:74–8.
- Voutsadakis IA, Doumas S, Tsapakidis K, Papagianni
M, Papandreou CN. Bone and brain metastases from ampullary
adenocarcinoma. World J Gastroenterol. 2009;15:2665–8.
- Malka D, Hammel P, Lévy P, Sauvanet A, Ruszniewski P, Belghiti
J, Bernades P. Splenic complications in chronic pancreatitis:
prevalence and risk factors in a medical-surgical series of 500
patients. Br J Surg. 1998;85:1645–9.
- Toussi HR, Cross KS, Sheehan SJ, Bouchier-Hayes D, Leahy AL. Spontaneous splenic rupture: a rare complication of acute pancreatitis. Br J Surg. 1996;83:632.
- Tseng CW, Chen CC, Chiang JH, Chang FY, Lin HC, Lee SD.
Percutaneous drainage of large subcapsular hematoma of the spleen
complicating acute pancreatitis. J Chin Med Assoc.
2008;71:92–5.
- Siu TL. Percutaneous drainage of spontaneous subcapsular
haematoma of the spleen complicating chronic pancreatitis.
Surgeon. 2004;2:52–5.