48uep6bbphidvals|430
48uep6bbph|2000F98CTab_Articles|Fulltext
Introduction
Ehlers-Danlos syndrome (EDS) is a connective tissue disorder
characterized by skin extensibility, abnormal wound healing
and joint hyper mobility.1-4 The striking clinical features are
hyper elastic smooth skin that is prone to splitting with minor
trauma, easy bruising, spontaneously resolving dislocations
of joints, scoliosis and ocular features such as keratoconus
depending on the type of EDS.1-8 Multiple hernias are usually
a rule rather than exception in these patients. However, the
abdominal symptoms are very rare presenting features of this
disease.
We have treated a boy suffering from EDS with multiple
hernias and poor wound healing. The unique feature in this
child was rectal redundancy that caused intestinal obstruction
and generalized abdominal distension, both of which got
resolved after partial resection of the redundant rectum. No
similar case was found in the literature.
Case Report
A one month old baby presented with right inguinal hernia, left
undescended testis with hernia and supraumbilical hernia. Ten
days following the repair of right inguinal hernia and
supraumbilical hernia, he presented with features of obstructed
left inguinal hernia for which a left inguinal herniotomy with
left orchidopexy (as he had left undescended testis as well)
was done. It was a large sliding hernia with appendix and a
floating caecum. Post-operatively, the baby developed
bronchiolitis that progressed to pneumonia and had to be
managed in pediatric ICU.
The baby developed scrotal wound dehiscence on the 7th
post-operative day, without any gross evidence of predisposing factors. However, with supportive management, the wound
healed with secondary intension. A week later the baby
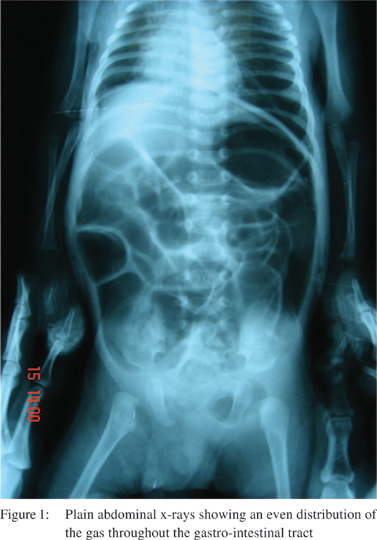
developed generalized abdominal distension. Plain abdominal
X-rays showed an even distribution of the gas throughout the gastro-intestinal tract up till rectum, with no cut off of gas
(Figure 1). There was no vomiting but the patient produced
high bilious aspirate through the nasogastric tube. Septic
screen was negative and bowel sounds were normal. Rectal
examination was normal and on direct questioning, the mother
gave past history of similar episodes of spontaneously
resolving abdominal distension. There was no history of similar
illness in the family. The distension decreased once the flatus
tube was passed rectally.
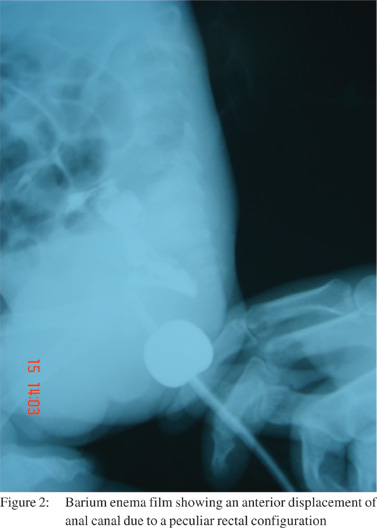
The barium enema study revealed no hold up of the contrast
in the sigmoid colon. On fluoroscopy, the rectum was found to have a large sac like profile with a prominent posterior bulge.
On carefully conducted pressure augmented barium enema, the anal canal appeared to have been anteriorly displaced due
to this peculiar rectal configuration (Figure 2). However, the most important observation on the lateral film was a straight
normal caliber anal canal and a Z-shaped rectum with sharp
bending curves. A provisional diagnosis of intestinal
obstruction due to a redundant rectum was considered. We
planned rectal trimming via the posterior sagittal route, an
approach that is commonly used in treating babies with
anorectal malformations worldwide.
This finding along with the other observation of multiple
hernias then drew our attention to the possibility of the presence of some connective tissue disorder. On examination of the baby,
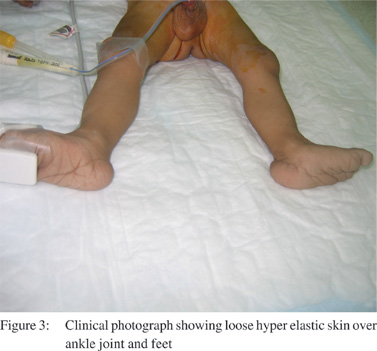
we found some other features consistent with EDS. They were
loose hyper elastic skin over the joints though joint hyper
mobility was not so clear (Figure 3). An earlier episode of delayed wound healing as well as large hernia could also be
explained now.
On exploration through the posterior sagittal route, the
rectum was found to be lengthy and redundant and could be
delivered into the wound easily; unlike a normal rectum which
lies snugly behind the bladder. There was no suggestion of a
mesorectum. About 3.5 cm length of the rectum was resected
at the point of maximum redundancy and a slightly oblique single layer anastomosis was carried out between the rectal
ends using 3-0 vicryl interrupted sutures. The pelvic floor muscles were adequately developed and they were sutured
with due precautions (since the baby had EDS) and protective colostomy was also done. The histopathological report of the
resected rectal segment showed mucosal congestion only. The neural elements were normal and thereby, the possibility of
Hirschsprung’s disease or neuronal intestinal dysplasia was
ruled out.
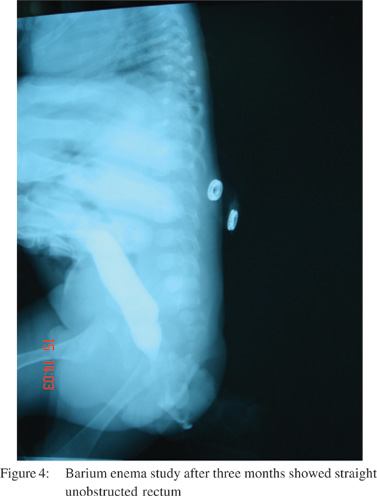
On 8th post-operative day the baby developed parastomal
wound dehiscence and bowel evisceration through this defect. Emergency repair was carried out for this. Barium enema study
after three months showed straight unobstructed rectum (Figure 4). Following this the colostomy stoma was closed.
Though the baby had been frequently requiring
hospitalizations for his medical condition including
gastroenteritis and bronchiolitis, his wounds have healed
completely and over past four years, he has not had any
problems in defecation
Discussion
EDS is a rare connective tissue disorder with an incidence of
1:5000 births.1 Different types (I - XI) have been described with certain type-specific clinical features,2-8 viz. type I (classic,
severe form) and type II (milder form) are characterized with
joint hypermobility, velvety skin, hyperextensibility and easy
scarring. The classic form is caused by defect in type V collagen due to defective COLA5A1 and COLA5A2 genes and
is inherited in an autosomal dominant pattern.[9] The basis of
the poor wound healing in these patients is the deficiency of
lysyl hydroxyl caused by gene mutation.[1]
The presentation pertaining to different systems differs in
different types. The facial features typically include widely
spaced eyes, high nasal bridge and prominent frontal
bossing.[6,7,8] While these facial features were not very prominent in the baby treated by us, the skin laxity especially around the
joints, feet and hands, was unmistakable and this together
with the features of multiple hernias and a delayed wound
healing clinched the diagnosis. The clinical features of the
baby treated by us classify his disorder under type 1 EDS.
We acknowledge a delay in reaching the diagnosis of EDS,
despite the presence of multiple hernias in our patient, owing
to rarity of this condition. It was the abnormally redundant
rectum in such a young child that finally caused us to suspect
a connective tissue disorder. Subsequently the other signs of
EDS were detected including multiple hernia, poor wound
healing and hyperelastic skin over the joints. The diagnosis of
EDS is mainly based on clinical features and a number of major
and minor clinical criteria, laid down by the medical advisory
group and the Ehler-Danlos Support Group, UK in 1997.[3] The
major criteria are: (a) skin hyperextensibity, (b) widened atrophic
scars, (c) joint hypermobility and (d) positive family history.
The combination of the first three major diagnostic criteria has
a high specificity for EDS, classic type. The presence of one or more minor criteria contributes to the diagnosis. The
biochemical tests are very expensive and generally not
employed for diagnostic purpose. The family history was not
positive in our patient, and joint hypermobility was not clear
as the child was quite young (the latter generally manifests
when a child starts to walk). However, other major and minor
criteria were present in the baby treated by us.
Among the various surgical conditions involving the rectosigmoid
region leading to constipation and intestinal
obstruction, Hirschsprung’s disease, neonatal intestinal
dysplasia, duplication cyst of the rectum and sigmoid volvulus
are prominent but to the best of our knowledge, no case of obstruction due to a redundant rectum has been reported till
date. The redundancy of the recto-sigmoid area in our patient
was in all probability due to the loose rectal connective tissue,its distal most location in the gut and thereby bearing the brunt
of gravity too.
Till date no case of EDS with a predominant abdominal
presentation and specifically with recurrent intestinal
obstruction due to a redundant rectum, has been reported,
though surgical complications of EDS have been described.[10]
In face of paucity of literature and our greater experience with
posterior sagittal anorectoplasty, it was considered worthwhile
to resect the redundant rectum through posterior sagittal
approach in order to straighten its course. Some authors do
advise supplementation of vitamin C in the patients with type
VI EDS, considering it to be caused by dysfunctional lysyl
hydroxyl enzyme.[1]
In short, although EDS is a rare condition, its possibility
should be kept in mind in a child presenting with multiple
hernias. The parents should also be screened for the signs of
this disease so that genetic counseling can be done for
subsequent pregnancies.
References
- Shashikiran U, Rastogi A, Gupta RP, Sabir M. Ehler-Danlos syndrome type VI variant presenting with recurrent respiratory
infections and responding to high dose vitamin C. J Assoc
Physicians India. 1999;47:554–5.
- Prockop DJ, Ala-kokko L. Inherited disorders of connective tissue.
In: Kasper DL, Braunwald E, Fauci AS, Hauser SL, Longo DL,
Jameson JL, editors. Harrison’s Principle of Internal Medicine.
New York: Mc Graw Hill Medical Publishing Division;
2005.p.2324–30.
- Beighton P, De Paepe A, Steinmann B, Tsipouras P, Wenstrup
RJ. Ehlers-Danlos Syndromes: revised nosology. Villefranche,
1997. Ehler- Danlos Foundation (USA) and Ehler-Danlos Support
Group (UK). Am J Med Genet. 1998;77:31–7.
- Yeowell NH, Pinnell SR: The Ehlers-Danlos Syndrome. Semin Dermatol. 993;12:229.
- Parikh F, Sivaramakrishnan A, Pai-dhunghat JV. Type VI Ehler Danlos Syndrome. J Assoc Physicians India. 2004;52:631.6.
- Pope FM, Narcisi P, Nicholls AC, Liberman M, Oorthuys JW.
Clinical presentations of Ehler Danlos Syndrome type IV. Arch
Dis Child. 1988;63:1016–25.
- Patel AB, Renge RL. Ehler-Danlos Syndrome. Indian Pediatr.
2002;39:784–5.
- Olaosebikan A, Wolf B. Ehlers-Danlos Syndrome in a Zimbabwean
child. Cent Afr J Med. 1993;39:20–2.
- Nicholls AC, Oliver JE, McCarron S, Harrison JB, Greenspan
DS, Pope FM. An exon skipping mutation of a type V collagen
gene (COL5A1) in Ehlers-Danlos syndrome. J Med Genet.
1996;33:940–6.
- McEntyre RL, Raffensperger JG. Surgical complications of Ehler-
Danlos syndrome in children. J Pediatr Surg. 1977;12:531–5.