|
Mithun Nariampalli Karthyarth1, Hemanga Kumar Bhattacharjee1, Ankur Goyal2, Saurabh Kedia3, Asit Ranjan Mridha4, Rajinder Parshad1 1Department of Surgical Disciplines, 2Department of Radio-diagnosis, 3Department of Gastroenterology and Human Nutrition, 4Department of Pathology, All India Institute of Medical Sciences, New Delhi, India.
Corresponding Author:
Dr Hemanga Kumar Bhattacharjee Email: dr_hkb75@yahoo.com
Abstract
Peutz-Jeghers Syndrome (PJS) is a rare inherited autosomal dominant disorder characterized by pigmented mucocutaneous melanotic macules and hamartomatous polyps. PJS arises due to mutations in STK11 gene located on chromosome 19q 13.3 and predisposes the patients to a multitude of malignancies with an estimated cumulative risk of 81% - 93%. Breast, gastrointestinal tract, pancreas, reproductive system and lung are common sites of development of malignancies in these patients. Anemia, rectal bleeding, abdominal pain, obstruction and intussusception are the usual complications in patients with PJS leading to multiple interventions. Upper GI endoscopy and Double Balloon Enteroscopy (DBE) allows screening of the gastrointestinal tract. Polypectomy of hamartomas more than 1 cm carried out at the time of surveillance endoscopy, abates the complications like bleeding, obstruction and intussusception. When DBE is not feasible, intraoperative endoscopy (IOE) is helpful to evaluate the entire gastrointestinal tract during surgery. IOE is also crucial for removal of all small intestinal polyps. Imaging techniques like magnetic resonance enterography and computed tomography enterography and video capsule endoscopy are non-invasive options for evaluation and screening in these patients. Sixty eight percent of the patients require emergency surgery during their lifetime. Regular cancer screening protocols should be instituted for early detection of malignancies. Genetic counseling and screening of other first degree family members helps in their preemptive identification and management. Chemoprevention using mTOR inhibitors, COX-2 inhibitors could be helpful in polyp reduction.
|
48uep6bbphidcol2|ID 48uep6bbphidvals|3036 48uep6bbph|2000F98CTab_Articles|Fulltext Introduction
Peutz-Jeghers syndrome (PJS) is a rare inherited disorder characterized by pigmented mucocutaneous melanotic macules and hamartomatous polyps in the gastrointestinal tract with varying degrees of cancer risk1,2,3. Jan Peutz first described this disorder in a Dutch family. Harold Jeghers subsequently described the relationship between pigmented lesions, gastrointestinal polyposis and increased risk of carcinoma4. The eponym of Peutz-Jeghers syndrome was coined in 1954. PJS is a rare entity. Recently, there is considerable advancement in its diagnosis and management. This review focuses on the diagnosis and management of PJS.
Genetics
PJS is caused by mutation in the STK11 gene located on the short arm of chromosome 19 (19p13.3). Most mutations are small deletions, insertions or single base substitutions leading to an abnormal truncated protein with no enzymatic activity1,3. The STK11 gene encodes a serine threonine kinase and acts as a tumor suppressor gene. Mutation in STK11 gene leads to activation of mammalian target of rapamycin (mTOR) pathway leading to increase in protein synthesis by phosphorylating and inhibiting the mRNA translational repressor proteins7,8,12. Inheritance follows an autosomal dominant pattern with variable penetrance resulting in differences in presentation among family members1,4,9. In 10%-20% patients PJS arises due to de novo mutation without any family history. It is noteworthy that STK11 mutation could be identified only in 30% - 70% of the patients10. Variation in techniques of mutation analysis and patient selection are the cause for low detection rates. Newer techniques of mutation analysis such as Multiplex ligation dependent probe amplification (MLPA) have increased the identification rates up to 94%. The possibility of other gene mutations causing PJS is also being probed11. Identification of mutation in mTOR pathway in causation PJS have led to use of mTOR inhibitors for chemoprevention in PJS.3
Clinical Features
The prevalence of PJS varies from 1 in 8300 to 1 in 2.8 lakh population with equal male to female preponderance1,3. The median age of onset of presentation varies from 11-13 years3. Hyper pigmented macules are commonest symptoms. These macules are seen commonly around the mouth, lips, eyes, nostrils, and on the buccal mucosa and sparsely on the fingers, soles of the feet, palms, anal area and intestinal mucosa1,2,3 (Figure 1, Figure 2). These are present in 95% of PJS patients. These arise due to clusters of melanocytes in the dermo-epidermal junction and have no malignant potential negating the need for biopsy. These hyper pigmentations may disappear by adolescence in many patients4,6.
Anemia, rectal bleeding, abdominal pain, obstruction and/or intussusception are the other common complications in patients with PJS in the first three decades.4,5 Ninety percent of affected individuals eventually develop small intestinal polyps13. In the small intestine, jejunum is the most common site for polyps followed by ileum and duodenum. Involvement of colon, stomach and rectum are seen in the decreasing order of incidence15. In a minority of patients, gallbladder, common bile duct, nasal mucosa, bladder, uterus and vagina2,15 are also affected by polyps. These polyps are hamartomatous polyps and characterized by complex glands with presence of arborizing smooth muscle bands in the lamina propria (arrows) on microscopy (Figure 3). The glands are lined by columnar epithelium and goblet cells primarily in the superficial portion and on the surface of the polyp. At the base, Paneth cells and endocrine cells may be seen.1,3
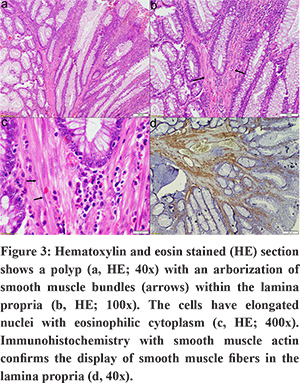
PJS is associated with increased risk of gastrointestinal and non gastrointestinal malignancies. The cumulative risk of development of malignancies is estimated between 81% - 93%.13,16. Breast cancer poses the highest specific risk affecting 32% - 54% of the females with PJS. Colon (39%) is the most common site of GI tract malignancy followed by pancreas (36%), stomach (29%), small intestine (13%) and esophagus13,19. Few cases of Cholangiocarcinoma and carcinoma of gallbladder are also reported3,21. Sex cord tumors of ovary is seen in 36% of female patients while sex-cord testicular tumors occur in 9% of male PJS patients13,15. PJS is associated with lung, uterine and cervical cancers with prevalence of 15%, 10% and 9% respectively. Surveillance and early detection hence play a crucial role in the management of PJS patients13,16.
Diagnosis and Treatment
In patients with a family history of PJS, diagnosis can be made with any number of PJS polyps, or characteristic prominent PJS mucocutaneous pigmentation20. The World health organization (WHO), Mayo Clinic, and Tomlinson and Houston have independently proposed guidelines for diagnosis of PJS (Table 1)17,20,22. Among these, WHO criteria are commonly used. It states that in patients without a family history of PJS, a diagnosis of PJS is made if there are three or more histologically confirmed PJS polyps, or any number of PJS polyps and characteristic PJS mucocutaneous pigmentation. In patients with a family history of PJS, diagnosis can be made with any number of PJS polyps, or characteristic prominent PJS mucocutaneous pigmentation20.

Genetic testing helps in confirming the diagnosis. STK11 mutation is identified in around 94% of clinically diagnosed patients11. Genetic testing should be carried out by 8 years of age in high risk first degree relatives of PJS patients without any clinical stigmata of PJS. First degree relatives of PJS patients with identified STK11 mutation should undergo testing for the specific mutation. Negative report for the mutation rules out PJS in the relatives, while a positive report confirms the disease and should follow the treatment protocol. If mutation could not be identified in the index case further testing of the relatives are futile and should be avoided1.
Imaging
Multi-detector Computer Tomography (MDCT), Magnetic resonance imaging (MRI) and Video capsule endoscopy (VCE) are used as non-invasive modalities for diagnosing PJS. Both CT and MR enterography have good sensitivity for larger polyps; however, the accuracy drops for sub-centimetric lesions.
Multi Detector Computed Tomography (MDCT)
MDCT enterography has high spatial resolution and is usually performed for the evaluation of small-bowel tumors and surveillance in patients with PJS. The polyps appear as well-defined hyper-dense enhancing lesions. These are usually multiple, often pedunculated lesions of varying size and more frequently involve the small bowel (Figure 4). CT enterography involves distending the small bowel loops that have neutral contrast enabling high lesion detection rates. The exposure to ionizing radiation however makes CECT a suboptimal candidate for repeat imaging which is needed for surveillance24,26. CT also shows polyp-related complications such as intussusception and bowel obstruction. Bowel within a bowel sign, target-like appearance or sausage shaped mass are imaging features of intussusception.
MR Imaging
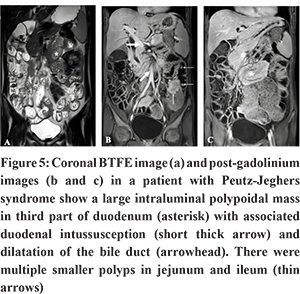
MR enterography is also based on a similar principle of optimally distending the small bowel loops to increase the sensitivity in the detection of polyps. MR however has better contrast resolution and provides multiple paradigms in the form of various pulse sequences to assess bowel polyps. The fluid used to distend the small bowel appears hyper intense on T2W images and hypointense on post-gadolinium images. Thus, luminal polyps appear dark inT2-weighted images and bright in post-contrast images (because they enhance) (Figure 5). The overall concordance rate between MR enterography and VCE is 70% and polyps measuring 15mm or higher have a better concordance rate of 93%. However, VCE has better rates of identification of polyps measuring 6mm to 9 mm. MR enterography appears to be a safe alternative to VCE for small bowel evaluation in PJS patients5,24. Being radiation-free, MR is a good method for follow-up and surveillance of these patients, especially since majority of these are young children and adults.
Video capsule endoscopy (VCE)
VCE have shown increased sensitivity in polyp detection when compared to conventional radiological techniques. VCE is a safe and diagnostically sensitive method for small bowel surveillance in patients with PJS including children. It causes substantially less patient discomfort than barium enterography or double balloon enteroscopy. VCE has been useful in diagnosing patients with occult gastrointestinal bleeding. VCE demonstrated higher polyp detection rate than small bowel follow-through in a blinded comparison in patients with PJ syndrome. In another study, the number of polyps >10 mm detected by VCE were greater than by MRE. However, the positive predictive value of detecting a polyp at subsequent balloon enteroscopy was lower with VCE than MRE (60% vs 100%). MRE was also more accurate in terms of size estimation and localization of polyps. In comparison with double balloon enteroscopy, VCE has a comparable polyp detection rate. Duodenal and proximal small intestinal polyps and lesions are missed in VCE due to lesser transit time. The other major drawback of VCE is the inability to accurately estimate polyp size and location. VCE is also contraindicated in patients presenting with obstruction33,34.
Endoscopy
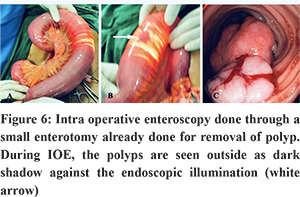
The two basic techniques in the diagnosis and management of hamartomas include Double balloon enteroscopy (DBE) and Intra-operative endoscopy (IOE)3,23. Double balloon enteroscopy was introduced as a modification of push enteroscopy by Yamamoto et al. in 2001 and has become the investigation of choice for evaluation of small bowel lesions and can evaluate the entire small bowel. It consists of an endoscope and a soft flexible over-tube, each having an inflatable balloon attached to its distal end27,29,30. Study comparing DBE with IOE showed similar polyp detection rates irrespective of size. In a comparative study of 18 consecutive patients, DBE demonstrated more polyps than small bowel follow-through and had comparable accuracy to VCE. Complications with DBE are rare with diagnostic procedures and 4.3% for therapeutic procedures. DBE however possesses a disadvantage of being a prolonged invasive procedure and in PJS patients its maneuverability is hampered as most patients had prior abdominal surgery3,5,24. Intraoperative endoscopy (IOE) is the combination of laparotomy or laparoscopy with endoscopy. During laparotomy or laparoscopy, an enterotomy is made on antimesenteric border on the small bowel and the endoscope is passed through it. Small bowel is inflated with air for better evaluation. It was the accepted method for complete evaluation of small bowel before the era of DBE. Also, large polyps obscuring the lumen will pose difficulty in DBE and mandate a conversion to IOE. Intraoperative endoscopy reduces the need for frequent enterotomies and also increases polyp detection rate.3,34,35 (Figure 6). Attempt should be made to clear all polyps in these patients utilizing IOE. Endoscopic therapy in the small intestine should be performed with special care to avoid complications such as bleeding and perforation5,23,35.
In a comparative study of DBE and IOE, both techniques had comparable accuracy in removing small bowel polyps and the authors concluded that polypectomy with DBE may obviate the need for repeated surgeries and DBE would be less invasive and convenient for the patient. Single Incision Laparoscopic (SILS) assisted DBE are currently being carried out in few centers and need further validation. It helps in easier conversion to IOE when DBE fails37. Spiral enteroscopy is a newer technique that uses a special over-tube called Discovery Small Bowel (DSB) to pleat small bowel. Spiral enteroscope consist of an overture with helical spirals of 5mm at the end39,40. This could be rotated independently from the enteroscope and can be engaged or disengaged when advancing. The technique of spiral enteroscopy is thus helpful in the diagnostic and therapeutic evaluation of small bowel by rotating and clockwise movement of enteroscope. However spiral enteroscopy needs further validation in polyp detection23,38,39. Virtual enteroscopy by use of MDCT technique is also being evaluated in the detection of polyps in PJS patients. Virtual enteroscopy has shown promising results in the detection of small bowel tumors40. Virtual enteroscopy involves inserting a duodenal tube under fluoroscopy followed by inflation of the small intestine with 2000ml of air. Air acts as a contrast medium to achieve small bowel expansion. This helps in better identification of raised lesions such as polyps compared to enterography40,41.
Screening
Screening for malignancy is pivotal in managing patients with PJS. For first degree relatives of PJS patients, screening should start at birth followed by annual history and physical examination. Special attention should be given to identification of mucocutaneous macules, precocious puberty and testicular tumors which appear at an early age.1,5 Multiple screening protocols have been elucidated by various institutions including Danish polyposis Registry, St Marks Hospital, Johns Hopkins Hospital, The Mayo Clinic for follow up of PJS patients3,5,42-47. Breast cancer poses the highest specific risk and screening in female PJS patients should be along the lines of screening of BRCA1 and BRCA2 patients. It includes monthly breast self-examination starting at 18 years of age and semiannual clinical breast examination and annual mammography starting at the age of 25 years. Baseline UGI endoscopy and colonoscopy have been suggested at 8 years of age. If polyps are present then colonoscopy and UGI endoscopy to be carried out 3 yearly. If no polyps were detected at the baseline investigation then reinitiate follow up at 18 years with colonoscopy and UGI endoscopy carried out every 3 years. Colonoscopy frequency to be increased after 50 years, conducted every 1-2 years. Pancreatic cancer is third most common cancer in PJS patients, however there are no evidence for routine screening protocols as cost benefit analysis showed poor cost effectiveness. Screening with pelvic examination and pap smear should be carried out yearly to rule out cervical and uterine malignancies starting at 21 years. The mean age of development of testicular cancer is 9 years prompting an annual examination from birth and attention to development of precocious puberty. Ultrasound examination to be done in case of any suspicious findings on examination.1,3,5,45-47 The surveillance protocol is outlined in Table 2.
Chemoprevention
PJS being a rare disease, chemopreventive human studies are difficult to conduct due to lack of participants49. Animal studies have shown promising results. Rapamycin is a macrolide antibiotic and acts as a mTOR inhibitor after binding to its cytoplasmic receptor3,12. Rapamycin studies in mice models showed reduction of polyp burden and polyp size48. Rapamycin analogues including sirolimus and everolimus approved for various cancer treatment could be helpful in PJS patients12. Another frontier in chemoprevention is the use of COX-2 inhibitors based on the observation that hamartomas and carcinomas in PJS patients expressed COX-2 receptors. Celecoxib, a COX-2 inhibitor, showed decreased vascularity and reduction in tumor burden in mice models50,51. Metformin, an oral hypoglycemic drug, has also shown inhibition of mTOR pathway in experimental models, and needs further validation51.
Surgery
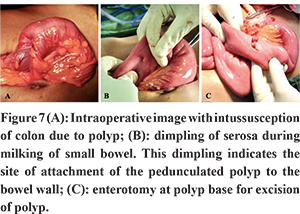
Laparotomy or laparoscopic surgery is the treatment for polyps related complications and for endoscopically irretrievable symptomatic polyps. About 30% of PJS patients required laparotomy before 10 years of age and by 20 years 68% of patients had undergone a laparotomy, mostly as an emergency procedure for complications as mentioned earlier3,14. Usually, the surgeon resorts to removal of the symptomatic polyp. However, in PJS patients the concept of “clean sweep surgery” in which all polyps are removed at the time of laparotomy have proven benefits by reducing need for subsequent laparotomies and increasing overall survival1,5,52. The clean sweep surgery could be carried out with the help of intraoperative endoscopy or by multiple enterotomies at centers where intraoperative endoscopy is unavailable. Laparoscopy, enterotomy and polyp excision is also a viable option in these patients46. Intraoperative milking of small bowel drags the polyp and produces dimpling on the bowel wall and can be used as an adjunct in identifying and locating the base of polyps. Enterotomy is performed at the site of the dimple and the poly is excised including its base (Figure 7). Duodenal polyps arising near the papilla favors towards surgical resection. Endoscopic snaring or cautery ligation can lead to injury of papilla and should be avoided. Mucosectomy and papilla reconstruction is a viable surgical option in these patients. Irreducible intussusception, large polyp, multiple polyps in a short segment of bowel, vascular compromise due to obstruction require segmental resection of the bowel. Repeated laparotomy with extensive small bowel resections can eventually lead to short-bowel syndrome in these patients. Mostly resections are done for intussusception, and this could be avoided by prophylactic polypectomy of polyps measuring more than 1 cm during surveillance enteroscopy or doing a clean sweep surgery during the primary laparotomy3,14,45,46.
Road Ahead
The major deterrent in the management of these patients is the lack of awareness among patients and medical fraternity delaying diagnosis and early referral. Lack of national and international databases for collection and compilation of data of rare disorders including PJS hamper the progress. Strict adherence to surveillance protocols will prolong the life expectancy in these patients. Genetic counseling should be mandated for all patients. A centralized organization at national and international level may be set up for disseminating information to patients and health professionals, testing, diagnosing, follow up and clinical research for all rare inherited disorders. All diagnosed patients should be counseled for participating in clinical trials to deduct better treatment protocols. The advent of novel Clustered Regularly Interspaced Palindromic Sequences (CRISPR) technology and CRISPR associated protein 9 (Cas 9) mediated genetic editing as a cure is promising for PJS patients. Editing the mutated STK11 gene offers a radical and permanent cure for PJS patients. Still in the embryonal stages of development gene editing could hold the future to treat these rare inherited disorders53-55.
References - Giardiello FM, Trimbath JD. Peutz-Jeghers syndrome and management recommendations. Clinical Gastroenterology and Hepatology. 2006;4(4):408-15.
- Vogel T, Schumacher V, Saleh A, Trojan J, Möslein G. Extraintestinal polyps in Peutz-Jeghers syndrome: presentation of four cases and review of the literature. International journal of colorectal disease. 2000;15(2):118-23.
- Kopacova M, Tacheci I, Rejchrt S, Bures J. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World journal of gastroenterology: WJG. 2009;15(43):5397
- Keller JJ, Offerhaus GJ, Giardiello FM, Menko FH. Jan Peutz, Harold Jeghers and a remarkable combination of polyposis and pigmentation of the skin and mucous membranes. Familial cancer. 2001;1(3-4):181-5.
- Beggs AD, Latchford AR, Vasen HF, Moslein G, Alonso A, Aretz S, Bertario L, Blanco I, Bülow S, Burn J, Capellá G. Peutz–Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59(7):975-86.
- Fraser JD, Briggs SE, Peter SD, De Petris G, Heppell J. Intussusception in the adult: an unsuspected case of Peutz–Jeghers syndrome with review of the literature. Familial cancer. 2009;8(2):95-101
- Amos CI, Keitheri-Cheteri MB, Sabripour M, Wei C, McGarrity TJ, Seldin MF, Nations L, Lynch PM, Fidder HH, Friedman E, Frazier ML. Genotype–phenotype correlations in Peutz-Jeghers syndrome. Journal of medical genetics. 2004;41(5):327-33
- Entius MM, Keller JJ, Westerman AM, Van Rees BP, van Velthuysen MF, de Goeij AF, Wilson JH, Giardiello FM, Offerhaus GJ. Molecular genetic alterations in hamartomatous polyps and carcinomas of patients with Peutz-Jeghers syndrome. Journal of clinical pathology. 2001;54(2):126-31
- Chen HM, Fang JY. Genetics of the hamartomatous polyposis syndromes: a molecular review. International journal of colorectal disease. 2009;24(8):865-74.
- Hearle N, Schumacher V, Menko FH, Olschwang S, Boardman LA, Gille JJ, Keller JJ, Westerman AM, Scott RJ, Lim W, Trimbath JD. STK11 status and intussusception risk in Peutz-Jeghers syndrome. Journal of medical genetics. 2006;43(8):e41
- Aretz S, Stienen D, Uhlhaas S, Loff S, Back W, Pagenstecher C, McLeod DR, Graham GE, Mangold E, Santer R, Propping P. High proportion of large genomic STK11 deletions in Peutz-Jeghers syndrome. Human mutation. 2005;26(6):513-9.
- Kuwada SK, Burt R. A rationale for mTOR inhibitors as chemoprevention agents in Peutz-Jeghers syndrome. Familial cancer. 2011;10(3):469.
- Gammon A, Jasperson K, Kohlmann W, Burt RW. Hamartomatous polyposis syndromes. Best Practice & Research Clinical Gastroenterology. 2009;23(2):219-31.
- Hinds R, Philp C, Hyer W, Fell JM. Complications of childhood Peutz-Jeghers syndrome: implications for pediatric screening. Journal of pediatric gastroenterology and nutrition. 2004;39(2):219-20.
- Utsunomiya J, Gocho H, Miyanaga T, Hamaguchi E, Kashimure A. Peutz-Jeghers syndrome: its natural course and management. The Johns Hopkins medical journal. 1975;136(2):71-82.
- Giardiello FM, Brensinger JD, Tersmette AC, Goodman SN, Petersen GM, Booker SV, Cruz–Correa M, Offerhaus JA. Very high risk of cancer in familial Peutz–Jeghers syndrome. Gastroenterology. 2000;119(6):1447-53.
- Shaco-Levy R, Jasperson KW, Martin K, Samadder NJ, Burt RW, Ying J, Bronner MP. Morphologic characterization of hamartomatous gastrointestinal polyps in Cowden syndrome, Peutz-Jeghers syndrome, and juvenile polyposis syndrome. Human pathology. 2016;49:39-48.
- Riegert-Johnson D, Roberts M, Gleeson FC, Krishna M, Boardman L. Case studies in the diagnosis and management of Peutz-Jeghers syndrome. Familial cancer. 2011;10(3):463-8.
- Mehenni H, Resta N, Park JG, Miyaki M, Guanti G, Costanza MC. Cancer risks in LKB1 germline mutation carriers. Gut. 2006 Jul 1;55(7):984-90
- Hamilton SR, Aaltonen LA, editors. Pathology and genetics of tumours of the digestive system. Lyon:: IARC press; 2000 Sep 20.
- Parker MC, Knight M. Peutz-Jeghers syndrome causing obstructive jaundice due to polyp in common bile duct. Journal of the Royal Society of Medicine. 1983;76(8):701-3.
- Tomlinson IP, Houlston RS. Peutz-Jeghers syndrome. Journal of Medical Genetics. 1997;34(12):1007-11.
- Tomas C, Soyer P, Dohan A, Dray X, Boudiaf M, Hoeffel C. Update on imaging of Peutz-Jeghers syndrome. World Journal of Gastroenterology: WJG. 2014;20(31):10864.
- Gupta A, Postgate AJ, Burling D, Ilangovan R, Marshall M, Phillips RK, Clark SK, Fraser CH. A prospective study of MR enterography versus capsule endoscopy for the surveillance of adult patients with Peutz-Jeghers syndrome. American journal of roentgenology. 2010;195(1):108-16.
- Maccioni F, Al Ansari N, Mazzamurro F, Barchetti F, Marini M. Surveillance of patients affected by Peutz-Jeghers syndrome: diagnostic value of MR enterography in prone and supine position. Abdominal imaging. 2012;37(2):279-87.
- Amzallag-Bellenger E, Oudjit A, Ruiz A, Cadiot G, Soyer PA, Hoeffel CC. Effectiveness of MR enterography for the assessment of small-bowel diseases beyond Crohn disease. Radiographics. 2012;32(5):1423-44.
- Soyer P, Aout M, Hoeffel C, Vicaut E, Placé V, Boudiaf M. Helical CT-enteroclysis in the detection of small-bowel tumours: a meta-analysis. European radiology. 2013;23(2):388-99
- Maglinte DD. Capsule imaging and the role of radiology in the investigation of diseases of the small bowel. Radiology. 2005;236(3):763-7.
- Soares J, Lopes L, Boas GV, Pinho C. Wireless capsule endoscopy for evaluation of phenotypic expression of small-bowel polyps in patients with Peutz-Jeghers syndrome and in symptomatic first-degree relatives. Endoscopy. 2004;36(12):1060-6.
- Yamamoto H, Sugano K. A new method of enteroscopy–the double-balloon method. Canadian Journal of Gastroenterology and Hepatology. 2003;17(4):273-4.
- Yano T, Yamamoto H. Current state of double balloon endoscopy: the latest approach to small intestinal diseases. Journal of gastroenterology and hepatology. 2009;24(2):185-92.
- Matsumoto Y, Manabe N, Tanaka S, Fukumoto A, Yamaguchi T, Shimamoto M, Nakao M, Mitsuoka Y, Chayama K. Small-intestinal Peutz-Jeghers polyps resected by endoscopic polypectomy with double-balloon enteroscopy and removal confirmed by ultrasonography. Digestive diseases and sciences. 2006;51(12):2337-40
- Fry LC, Neumann H, Kuester D, Kuhn R, Bellutti M, Malfertheiner P, Monkemuller K. Small bowel polyps and tumours: endoscopic detection and treatment by double-balloon enteroscopy. Alimentary pharmacology & therapeutics. 2009;29(1):135-42.
- Kopácová M, Bureš J, Vykouril L, Hladík P, Šimkovic D, Jon B, Ferko A, Tachecí I, Rejchrt S. Intraoperative endoscopy. Surgical endoscopy. 2007;21(7):1111-6
- Schulz HJ, Schmidt H. Intraoperative endoscopy. Gastrointestinal Endoscopy Clinics. 2009;19(3):371-9.
- Zanoni EC, Averbach M, Borges JL, Corrêa PA, Cutait R. Laparoscopic treatment of intestinal intussusception in the Peutz-Jeghers syndrome: case report and review of the literature. Surgical Laparoscopy Endoscopy & Percutaneous Techniques. 2003;13(4):280-2.
- Stasinos I, Kamperidis N, Murino A, Jenkins JT, Warusavitarne J, Fraser C, Humphries A. Single incision laparoscopic assisted double balloon enteroscopy: a novel technique to manage small bowel pathology. Surgical Endoscopy. 2020;18:1-7.
- Frieling T, Heise J, Sassenrath W, Hülsdonk A, Kreysel C. Prospective comparison between double-balloon enteroscopy and spiral enteroscopy. Endoscopy. 2010;42(11):885-8.
- Akerman PA, Agrawal D, Chen W, Cantero D, Avila J, Pangtay J. Spiral enteroscopy: a novel method of enteroscopy by using the Endo-Ease Discovery SB overtube and a pediatric colonoscope. Gastrointestinal endoscopy. 2009;69(2):327-32
- Endo K, Utano K, Togashi K, Yano T, Lefor AT, Yamamoto H, Yasuda Y, Sugimoto H. Virtual enteroscopy using air as the contrast material: a preliminary feasibility study. Digestive Endoscopy. 2010;22(3):205-10.
- Haneda R, Sato S, Ohno K, Yoshikawa T, Takagi M. Usefulness of virtual enteroscopy for the detection of small polypoid lesion in the small bowel, a case report. International Journal of Surgery Case Reports. 2020;67:5-8.
- Zbuk KM, Eng C. Hamartomatous polyposis syndromes. Nature clinical practice Gastroenterology & hepatology. 2007;4(9):492-502
- Jelsig AM, Qvist N, Brusgaard K, Nielsen CB, Hansen TP, Ousager LB. Hamartomatous polyposis syndromes: a review. Orphanet journal of rare diseases. 2014;9(1):101.
- Pedersen IR, Hartvigsen A, Hansen BF, Toftgaard C, Konstantin-Hansen K, Büllow S. Management of Peutz-Jeghers syndrome. International journal of colorectal disease. 1994;9(4):177-9.
- Dunlop MG. Guidance on gastrointestinal surveillance for hereditary non-polyposis colorectal cancer, familial adenomatous polypolis, juvenile polyposis, and Peutz-Jeghers syndrome. Gut. 2002;51(suppl 5):v21-7.
- Armstrong D, Bacon J, Booker SV, Brensinger J, Chen P, Codori AM, Giardiello F, Grinnin CA, Johnson KA, Kim JW, Olsen S. The Johns Hopkins guide for patients and families: Peutz-Jeghers syndrome. The Johns Hopkins University. 2001.
- Wei C, Amos CI, Zhang N, Zhu J, Wang X, Frazier ML. Chemopreventive efficacy of rapamycin on Peutz–Jeghers syndrome in a mouse model. Cancer letters. 2009;277(2):149-54.
- De Brabander J, Eskens FA, Korsse SE, Dekker E, Dewint P, van Leerdam ME, van Eeden S, Klümpen HJ. Chemoprevention in Patients with Peutz-Jeghers Syndrome: Lessons Learned. The oncologist. 2018;23(4):399.
- Rossi DJ, Ylikorkala A, Korsisaari N, Salovaara R, Luukko K, Launonen V, Henkemeyer M, Ristimäki A, Aaltonen LA, Mäkelä TP. Induction of cyclooxygenase-2 in a mouse model of Peutz–Jeghers polyposis. Proceedings of the National Academy of Sciences. 2002;99(19):12327-32
- McGarrity TJ, Peiffer LP, Amos CI, Frazier ML, Ward MG, Howett MK. Overexpression of cyclooxygenase 2 in hamartomatous polyps of Peutz-Jeghers syndrome. The American journal of gastroenterology. 2003;98(3):671-8.
- Dowling RJ, Zakikhani M, Fantus IG, Pollak M, Sonenberg N. Metformin inhibits mammalian target of rapamycin–dependent translation initiation in breast cancer cells. Cancer research. 2007;67(22):10804-12.
- Oncel M, Remzi FH, Church JM, Connor JT, Fazio VW. Benefits of ‘clean sweep’in Peutz-Jeghers patients. Colorectal Disease. 2004;6(5):332-5.
- Tang L, Zeng Y, Du H, Gong M, Peng J, Zhang B, Lei M, Zhao F, Wang W, Li X, Liu J. CRISPR/Cas9-mediated gene editing in human zygotes using Cas9 protein. Molecular genetics and genomics. 2017;292(3):525-33.
- Lee K, Conboy M, Park HM, Jiang F, Kim HJ, Dewitt MA, Mackley VA, Chang K, Rao A, Skinner C, Shobha T. Nanoparticle delivery of Cas9 ribonucleoprotein and donor DNA in vivo induces homology-directed DNA repair. Nature biomedical engineering. 2017;1(11):889.
- Tabebordbar M, Zhu K, Cheng JK, Chew WL, Widrick JJ, Yan WX, Maesner C, Wu EY, Xiao R, Ran FA, Cong L. In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science. 2016;351(6271):407-11.
|