48uep6bbphidcol2|ID
48uep6bbphidvals|2967
48uep6bbph|2000F98CTab_Articles|Fulltext
Introduction
Chronic pancreatitis (CP) is a chronic inflammatory condition characterized by fibrosis of the gland. It clinically manifests with severe abdominal pain, exocrine and endocrine insufficiency. Although there are many mechanisms of pain in CP, the most accepted one is increased intraductal/intraparenchymal pressure, known as‘ theplumbing theory’, which forms the theoretical background of most interventions including surgical and endoscopic drainage procedures1. Several studies have suggested that surgical procedures are superior to and more effective than endoscopic therapy for management of CP2,3. Surgical procedures for CP are divided into resectional procedures, drainage procedures and a combination of both of these. Lateral pancreatojejunostomy (LPJ) is the commonly done drainage procedure. It is traditionally done as an open procedure. With advancement in laparoscopic surgery, laparoscopic LPJ is recently gaining acceptance in many centers. However, many studies are limited to case reports/case series. Our center isa tertiary care referral center for hepato-pancreato-biliary (HPB) diseases with a dedicated and experienced team of laparoscopic surgeons. We have earlier published our experience of managing HPB diseases including malignancies laparoscopically4,5. Hereby we would like to share our experience of laparoscopic LPJ in the management of CP and to the best of our knowledge; it is one of the largest series in the world.
Patients and Methods
We did a retrospective analysis of a prospectively maintained patient database of chronic pancreatitis patients undergoing surgical intervention in the department of surgical gastroenterology, GB Pant institute of Post Graduate Medical Education and Research, New Delhi from January 2007 to April 2018.
Of more than 450 patients who underwent surgical intervention for CP during the study period, 41 patients were treated by laparoscopic LPJ which constituted the study group. Before subjecting the patients to surgery, their need for surgery and the expected outcomes were discussed in a multidisciplinary meeting consisting of surgeons, medical gastroenterologists and radiologists. Once the decision for surgery was undertaken, all patients were thoroughly evaluated by history and physical examination. Routine blood investigations including hemogram, liver function tests (LFT), kidney function tests (KFT), serum electrolytes (SE), blood sugar, chest X ray and electrocardiogram (ECG) were done in all the patients. Blood sugar levels were optimised in diabetic patients and HbA1c was done before surgery. Tumour markers (carcino-embryonic antigen CEA and CA 19-9)were done in patients suspected to have a mass in the pancreatic head.
Radiological investigations performed in patients with suspected pancreatic head mass included ultra-sonography (USG), contrast enhanced computed tomography (CECT) (Figure 1a and 1b), magnetic resonance imaging with cholangio-pancreatography (MRCP) (Figure 1c and 1d), and endoscopic ultrasound (EUS) with or without fine needle aspiration cytology (FNAC).
Surgical Technique
After induction by general anesthesia, nasogastric tube and Foleys catheter were inserted. Patients were placed in supine leg split position with 45 degree elevation. Pneumoperitoneum was created by Veress needle through Palmer’s point.
Port positioning
11 mm camera port- 3 cm, infra umbilical in the midline.
5 mm ports- right and left para rectal, above and lateral to camera port.
5 mm port- epigastric.
12 mm port- right iliac fossa in the midclavicular line.
Exposure of the pancreas
The lesser sac was entered by dividing the gastro-colic omentum away from the gastro-epiploic arcade (Figure 2a). Adhesions between the pancreas and posterior surface of the stomach were released if present. Right gastro epiploic vessels were ligated if necessary. The entire anterior surface of the pancreas from C-loop of the duodenum to the tail of the pancreas was exposed.
Lifting the stomach away from the pancreas
Nylon suture 2-0 was passed from the left side of the abdominal wall, two sero-muscular bites were taken from the posterior surface of the stomach and then brought out through the abdominal wall on the right side. Both ends are tied on the abdominal wall lifting the stomach away from the pancreatic surface.
Identifying and opening the pancreatic duct
In patients with dilated pancreatic duct, studded with multiple large stones, the duct was opened directly using a harmonic scalpel keeping the active limb directly over the stone. In other patients, needle puncture was done to confirm the site of duct (Figure 2b) and then the puncture site was enlarged using the harmonic scalpel (Figure 2c).The duct was opened on both the sides, extending from tail of the pancreas to within 1cm of C-loop of duodenum (Figure 2d). Stones were removed by using Maryland dissector and endo-bowel grasper (Figure 3a) and were collected in an endobag and removed. (Figure 3b)
Creating the Roux limb of jejunum
The jejunal limb was divided 20 cm distal to duodeno-jejunal (DJ) flexure by using endo GIA stapler. The Roux limb was taken to the supracolic compartment through the mesocolic window and fixed to the tail of the pancreas. Side to side jejuno-jejunal (J-J) anastomosis was done by using endo GIA stapler (Figure 3c), and the common enterotomy site was closed by using polypropelene 3-0, in single layer continuous fashion.
Lateral pancreatico-jejunosotomy (LPJ)
The jejunal limb was opened in the ante-mesenteric border using harmonic scalpel. LPJ was done by using PDS 3-0, in continuous fashion, starting from the tail end, initially in the posterior layer, and then continued anteriorly at the head region towards the tail of the pancreas (Figure 3d).
Drain placement
After ensuring haemostasis, drain was placed into the lesser sac.
The port sites (>10 mm) sheaths were closed using Vicryl No1 suture and the skin was closed with staples (Figure 4).
Patients were allowed orally on post operative day (POD) 1. The drain was removed when the output was less than 50 ml.
Follow up was done 1week after discharge and 1month after discharge followed by once every 3 months.
Results
During the study period, 41 patients underwent a laparoscopic LPJ and constituted the study group. Patients who were converted to open procedure and lap assisted procedures were excluded from the study. There were 26 males and 15 females. The mean age of the subjects was 30.7 (7-51) years. 2 patients were children (<12 years).Pain was the main indication of surgery in all the patients. Nine patients had diabetes and 6 patients had steatorrhea. None of the patients had gastric outlet obstruction or gastro-intestinal bleeding. The mean MPD diameter was 13.6 (8-25) mm. Almost all patients had pancreatic ductal calculi with or without parenchymal calcifications. The pancreatic duct was opened from head to tail of the pancreas, removing all the stones from the MPD and dilated side branches if any.
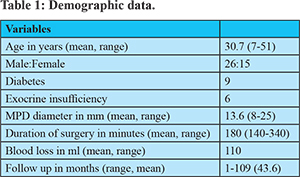
The mean duration of surgery was 180 minutes (140-340). The duration of surgery was more for the initial few cases and gradually decreased as the experience increased. In addition the surgical duration was also determined by the presence of peri-pancreatic adhesions and difficulty in identifying MPD in patients with small-sized ducts.However, with proper patient selection like absence of inflammatory changes and dilated MPD on preoperative imaging, the surgical duration could be reduced in a good number of patients.There was no procedure related mortality and no major complications. None of the patients developed pancreatic leak. Postoperative hospital stay was 5 (3-9) days and satisfactory pain relief was seen in 91% of patients with mean follow up of 43 months.
Discussion
Chronic pancreatitis (CP) is a progressive inflammatory disorder characterized by irreversible destruction of pancreatic parenchyma, associated with disabling chronic pain and permanent loss of exocrine and endocrine function6. Although there is limited understanding of the pathophysiological process of pain, ductal decompression remains the most effective and commonly done procedure in patients with dilated MPD and intractable pain. Surgical intervention for chronic pancreatitis is commonly accepted as the most effective therapeutic option for pain control and management of complications. When compared to endotherapy, surgery is associated with superior long-term pain reduction in patients with painful obstructive chronic pancreatitis. Surgical procedures are commonly divided into decompressive and resectional procedures or a combination of both. There is some evidence that early surgical duct decompression in patients with a dilated pancreatic duct andrelatively preserved glandular function can delay the onset of pancreatic exocrine and endocrine failure.
Lateral Roux-en-Y pancreatojejunostomy (Partington–Rochelle’s technique) is the most commonly done surgical decompression procedure, especially in patients with dilated pancreatic duct and without head mass. Traditionally, the procedure is done as an open technique.
As the exposure in laparoscopic procedures increases, the advantage of minimal access surgery can be extended to CP in selected group of patients. There has been a slow and steady rise in the application of minimally invasive surgery to pancreatic surgeries as is evident by the increasing number of laparoscopic pancreaticodudoenectomy series being published. However, there have been fewer series of laparoscopic LPJ.
Technical difficulty in controlling hemorrhage in the pancreatic region, dissection around important blood vessels, difficulty in identification of the duct, especially in the absence of laparoscopic ultrasound and the complexity involved in reconstruction have necessitated a steep learning curve for surgeons. Less bleeding in laparoscopic surgery has been attributed to magnification and better visualization. Few case series have concluded that laparoscopic LPJ being safe, feasible and effective in selected patients with CP. However, the basic requirement for the same is mastery in the technique of intra corporeal knotting and suturing8.
Kurian and Gagner were the first to perform successful laparoscopic pancreaticojejunostomy. In their study, four of the five patients were pain-free, with 5- to 30-month follow-up. They concluded that laparoscopic pancreaticojejunostomy can be performed safely in selected patients with chronic pancreatitis9.
In patients without a pancreatic head mass, both Frey’s and LPJ are equally efficacious in pain relief10. Although laparoscopic Frey’s procedure for CP is feasible in carefully selected patients, the risk of bleeding is more compared to simple drainage procedure leading to increased rates of conversion to open procedure11. In a study comparing LPJ and Frey’s procedure, both the procedures had equivalent benefit in terms of short-term pain reduction in selected patients. The authors also concluded that decompression of the pancreatic duct carried completely into the pancreatic head is a critical component in the success of the procedure10. All our patients underwent ductal decompression throughout the length of the gland as well from within 1cm of the ‘C’ loop of the duodenum to the tail of the pancreas. In our study, we excluded patients with suspected pancreatic head mass on preoperative imaging. We believe that, in patients without pancreatic head mass, head coring is not required, and ductal drainage alone is sufficient. Ductal drainage also helps decrease the morbidity associated with Frey’s procedure.
Another important factor to be considered while selecting patients for LLPJ is the diameter of the MPD, as it is technically easier to do intracorporeal suturing with larger duct as compared to small duct disease. The mean MPD diameter in our study was 13.6 mm with the smallest diameter being 8 mm. Identification of the pancreatic duct itself is an important part of the procedure, as few of our patients in the initial part of the study needed conversion to open procedure due to difficulty in identifying the duct.In patients with dilated pancreatic duct, studded with multiple large stones, the duct can be opened directly with a harmonic scalpel, keeping the active limb directly over the stone. In other patients, needle puncture and aspiration can be done to confirm the site of duct followed by enlarging the puncture site using the harmonic scalpel. Duct opening extending from the tail of the pancreas to within 1cm of the C-loop of the duodenum is critical as inadequate opening can lead to recurrence of pain. Stones which are present in the side branches should also be removed. Stones were removed using Maryland dissector and endo-bowel grasper and were collected in an endobag.
In the preoperative workup, it is essential to exclude the presence of any pancreatic and peripancreatic inflammatory changes as they may be associated with adhesions between the pancreas and the posterior gastric wall. In addition there may be excessive bleeding and difficulty in identifying the duct.
Mastery in intracorporeal suturing and knotting is an essential component of LLPJ. We did pancreatico-jejunal anastomosis by both interrupted and continuous suture technique. Although both the techniques are equal in terms of pancreatic leak, continuous suturing is less time consuming and bleeding from the cut edges of the duct and jejunum, if any, is better controlled. Single layer anastomosis was done using 3-0 PDS suture,starting from the tail end of the pancreas posteriorly and continuing into the anterior part at the head region.
Laparoscopic LPJ is an effective decompressive procedure for pain relief in patients with chronic calcific pancreatitis with ductal dilatation with the added advantages of minimal invasive surgery like cosmesis, less pain and a short hospital stay. The mean hospital stay was 5 days, which is shorter than that of open pancreaticojejunostomy (median hospital stayof 8 days)12. Similar to adults, LLPJ for children with chronic pancreatitis is safe and effective. In a study by Jin-Shan Zhang et al. 4 patients with chronic pancreatitis underwent LLPJ, with operative times ranging from 103 to 154 min. The averagepostoperative hospital staywas 4 to 6 days13. However, all the patients had protein plugs causing ductal obstruction. In our study, 2 patients were children (<12 years) and both had dilated ducts with multiple calculi.Mean follow up was 43.6 months (ranging from 1-109 months).
In the initial few cases, laparoscopic assisted LPJ was done with the open part involving mainly the LPJ anastomosis. However, with increase in experience we were able to complete the procedure laparoscopically with simultaneous improvement in the operating time. Few cases also needed conversion to open procedure due to dense adhesions and bleeding. However, both laparoscopic assisted and laparoscopic converted procedures were excluded from the study.
The successful outcome of LLPJ is mainly based on the selection of patients preoperatively. To summarize, dilated ducts without head mass and inflammatory changes are the key to success of the surgery and better post-operative outcomes. Cross sectional imaging, especially CECT abdomen is the main imaging modality that helps us in the proper selection of these patients.
Conclusions
Laparoscopic LPJ is feasible and safe with good short and long-term outcomes in selected groups of patients. However, it is a technically demanding procedure and should be done by a surgeon well versed with laparoscopic skills including suturing and knotting.
References
- LIEB II JG, Forsmark CE. Pain and chronic pancreatitis. Alimentary pharmacology & therapeutics. 2009 1;29:706-19.
- Dite P, Ružicka M, Zboril V, Novotný I. A prospective, randomized trial comparing endoscopic and surgical therapy for chronic pancreatitis. Endoscopy. 2003;35:553-8.
- Cahen DL, Gouma DJ, Yung N, Rauws EAJ, Boermeester MA, Busch OR, et al. Endoscopic versus surgical drainage of the pancreatic duct in chronic pancreatitis. N Engl J Med 2007; 356:676-84. [PMID: 17301298]
- Hepatic hydatid: PAIR, drain or resect? Gupta N, Javed A, Puri S, Jain S, Singh S, Agarwal AK. J Gastrointest Surg (2011) 15:1829–36
- Agarwal AK, Javed A, Kalayarasan R, Sakhuja P. Minimally invasive versus the conventional open surgical approach of a radical cholecystectomy for gallbladder cancer: a retrospective comparative study. HPB. 2015;17:536-41.
- Ahmed SA, Wray C, Rilo HL, Choe KA, Gelrud A, Howington JA, Lowy AM, Matthews JB. Chronic pancreatitis: recent advances and ongoing challenges. Current problems in surgery. 2006;43:127-238.
- Nealon WH, Thompson JC. Progressive loss of pancreatic function in chronic pancreatitis is delayed by main pancreatic duct decompression. A longitudinal prospective analysis of the modified puestow procedure. Annals of surgery. 1993 ;217:458.
- Palanivelu C, Shetty R, Jani K, Rajan PS, Sendhilkumar K, Parthasarthi R, Malladi V. Laparoscopic lateral pancreaticojejunostomy. Surgical Endoscopy And Other Interventional Techniques. 2006;20:458-61.
- Kurian MS, Gagner M. Laparoscopic side-to-side pancreaticojejunostomy (Partington-Rochelle) for chronic pancreatitis. Journal of hepato-biliary-pancreatic surgery. 1999;6:382-6.
- Terrace JD, Paterson HM, Garden OJ, Parks RW, Madhavan KK. Results of decompression surgery for pain in chronic pancreatitis. HPB. 2007;9:308-11.
- Tan CL, Zhang H, Li KZ. Single center experience in selecting the laparoscopic Frey procedure for chronic pancreatitis. World journal of gastroenterology. 2015;21:12644.
- Chiu B, Lopoo J, Superina RA. Longitudinal pancreaticojejunostomy and selective biliary diversion for chronic pancreatitis in children. Journal of pediatric surgery. 2006 1;41:946-9.
- Zhang JS, Li L, Liu SL, Hou WY, Diao M, Zhang J, Li SL, Ming AX, Liu Y, Wang HB, Cheng W. Laparoscopic pancreaticojejunostomy for pancreatic ductal dilatation in children. Journal of pediatric surgery. 2012;47:2349-52.