48uep6bbphidcol2|ID
48uep6bbphidvals|3050
48uep6bbph|2000F98CTab_Articles|Fulltext
Introduction
Amoebic liver abscesses (ALA) accounts for 3-9% of all cases of amoebiasis1. The high prevalence of infection in India and other tropical and subtropical regions of the world is attributed to poor education, poverty, overcrowding, contaminated water supply, and unsanitary conditionsetc.2 Alcohol is commonly believed to play an important role in the aetiology of ALA and ratesof alcohol intake in patients suffering from ALA vary from 20% - 62.5%.1,3 Heavy ethanol consumption (> 150 gm per day) produces a wide spectrum of hepatic lesions, the most characteristic being fatty liver (i.e., steatosis), hepatitis, and fibrosis/cirrhosis4. Steatosis is the earliest and the most common response that develops in over 90 per cent of problem drinkers who consume 4 to 5 standard drinks per day over decades. It is characterized by the deposition of fat in the hepatocytes and is a reversible condition. However, patients with chronic steatosis are more susceptible to fibrotic liver disease. Alcoholic hepatitis is a more severe, inflammatory type of liver injury characterized by swollen, dying hepatocytes. Fibrosis and its terminal or late stage, cirrhosis, refer to the deposition of abnormal amounts of extracellular matrix proteins. Patients initially exhibit active pericellular fibrosis, which may progress to cirrhosis, the late stage of hepatic scarring.
Despite a high prevalence, the diagnosis of ALD remains a dilemma. Clinically patients with ALD may exhibit the classical signs of liver disease including gynecomastia, Dupuytren’s contracture, parotid gland enlargement, dilated abdominal wall veins, and spider angiomas. However, these features manifest only when the disease is much advanced. Further patients with only fatty liver are usually asymptomatic and maintain an intact synthetic hepatic function5. Hence for the initial evaluation of suspected ALD, several questionnaires and laboratory tests areavailable to screen for alcohol intake and can be used5. However, liver biopsy remains the gold-standard diagnostic tool for ALD5.
ALA in the background of ALD has been reported to have a more fulminant course as patients are prone to develop acute on chronic liver failure6. However, no specific management protocols have been defined for this group of patients. Hence the first step in this direction would be to study how the clinical course and outcomeof treatment of ALA withALD differ from thosepatients without ALD. This will help in identifying patients at higher risk so that appropriate management protocols can be defined. Hence this prospective observational study was planned with the following objectives: 1) to study the severity of infection and rate of complications in patients of ALA with ALD, as compared to those without ALD; 2) to compare the response to medical treatment and indications for surgical interventions; 3) to compare the length of hospital stay and mortality rates between the two groups of patients
Materials and Methods
This longitudinal observation study was conducted on patients of ALA who presented to the department of surgery, UCMS & GTB Hospital, Delhi from a period of November 2018 to April 2020. Institutional ethics committee clearance was obtained for the conduct of the study and informed consent was taken from all the patients before inclusion in the study. Patient recruitment followed the non-probability convenience methodof sampling. A sample size of 120 patients was taken, out of which there were 60 consecutive patients of ALA without ALD (Group-I) and 60 consecutive patients of ALA with ALD (Group II). All these patients were admitted under the care of a single surgical unit.
Inclusion criteria followed were patients more than 18 years of age admitted with the diagnosis of amoebic liver abscess. A diagnosis of ALA was made on the criteria of clinical symptoms (pain upper abdomen, fever, hepatomegaly), laboratory investigations (leucocytosis, deranged liver functions), ultrasonography findings (suggesting of amoebic liver abscess), and aspiration (whenever indicated) of anchovy sauce pus and culture showing no bacterial growth7. Exclusion criteria were: patients with pyogenic liver abscess ( diagnosis based on clinical symptoms like high-grade fever, and raised leucocyte counts and pus culture showing bacterial growth wherever indicated) and patientswith serious co-morbidities such as diabetes, CAD, COPD and those on medications like steroids and patients of known chronic liver disease due to aetiologies other than ALD.
Diagnosis of ALD was made based on the history of significant alcohol intake, abnormality in liver profiles with serologic evidence (deranged liver enzymes, INR =1.5, albumin =3mg/dl) and/or radiological evidence of cirrhosis5.“Alcoholism” was screened as per the CAGE questionnaire. Depending on the frequency of alcohol intake, patients were divided into non-drinkers, occasional drinkers (alcohol intake < 3 times/week), and regular drinkers (alcohol intake = 3 times/week)1,8.
Demographic and clinical details of the patients admitted with a diagnosis of ALA were collected and entered in predesigned proforma. Their clinical course was carefully monitored for resolution of symptoms, change in laboratory parameters, development of complications like jaundice, rupture into pleural or peritoneal cavity, acute on chronic liver failure etc. For management of liver abscesses, four treatment categories were made comprising of (i) Conservative management, (ii) Percutaneous aspiration (PNA), (iii) Percutaneous drainage (PCD) and (iv) Open surgery. Patients with abscess size < 200 ml were treated conservatively, those between 200 ml to 400 ml were treated with PNA, while for abscess size >400 ml, PCD was placed.For the patient with a ruptured liver abscess causing peritonitis, open surgery was the modality of treatment. Patients showing signs of liver failure (jaundice, ascites, pedal oedema, hepatic encephalopathy or deranged INR) were treated with PCD even when abscess size was smaller (= 250 ml).
Acute on chronic liver failure ( ACLF) was defined as “acute hepatic insult manifesting as jaundice (serum bilirubin = 5 mg/dL) and coagulopathy (INR = 1.5), complicated within 4 weeks by ascites and/or encephalopathy in patients with previously diagnosed or undiagnosed chronic liver disease or cirrhosis6. Those patients who showed signs of acute hepatic injury leading to decompensation manifesting as coagulopathy and development of pedal oedema or ascites, but not meeting the criteria of ACLF were designated as patients with “Impending liver failure”. Patients with overt or impending liver failure received infusion of human albumin (5%)to tide over the period of acute decompensation in liver functions.
Antibiotics that were used as first-line treatment in our study were ciprofloxacin (400 mg twice a day intravenous or 500 mg twice a day orally) and metronidazole (1 gram intravenous or 800 mg orally thrice a day). Two weeks of metronidazole therapy was followed by 10 days of oral diloxanide furoate 500 mg three times a day. Higher antibiotics liketazobactam 4.5 mg thrice daily intravenously, orlinezolid 600 mg twice a day per oral,along with Metronidazole was started initially if patients had signs of decompensation like jaundice, ascites and pedal oedema, or if patients failed to respond within 48-72 hours, as noted by fever, increase in TLC or increasing abscess size. Patients who developed ACLF were started on albumin infusion (5% albumin) 100 mlonce daily for 3 to 5 days depending upon the resolution of oedema9.
The primary outcomemeasures were:1) estimates of the severity of disease based on laboratory parameters, mean size and number of abscesses;2) frequency of different complication 3) frequencies of use of different modalities of treatment; 4) mortality ; and 5) length of hospital stay in the two groups.
Statistical Analysis
Simple descriptive statistical analysis was used for the calculation of frequencies of various parameters. For comparison between the two groups, unpaired student’s t-test was used for quantitative variables and chi-square test was used for qualitative parameters.
Result
In Group I the mean age of the patients was 34.05±13.98 years, while in Group II it was 47.22 ±13.29 years. Thus, patients in group II were significantly older than group I.The overall male:female ratio in the study was (14:1) (M:F, 112:8). In Group I the male: female ratio was 9:1 whereas in Group II itwas 27:1.
Alcohol Consumption
In Group I, 32 patients gave a positive history for intake of alcohol whilein Group II, all 60 patients gave a positive history for intake of alcohol. The frequency of alcohol intake was divided into two categories, one with <3 times per week and other >3 times per week. In Group I out of 32 alcohol consumers, 19 consumed <3 times a week and 13 consumed >3 times a week. In Group II out of 60 patients 8consumed alcohol <3 times a week and 52 consumed >3 times a week. Years of Alcohol intake: The mean intake of alcohol in group I was 2.3± 2.5 years and in Group II was 8.37 ± 2.99 years. Hence, patients in group II had a history of significantly higher and longer duration of alcohol consumption. (Table 1)
Clinical Profile
Pain in right hypochondrium and fever were the most common symptoms in both groups. However, there was a significant difference between the groups as regards the prevalence of nausea, vomiting and fatigue. Signs like jaundice, splenomegaly, ascites and pedal oedema were also significantly different, as patients of ALA with ALD suffered from more severe outcomes. The mean size of the abscess cavity was also significantly larger in patients with ALD (391.20 ± 208.7 cc in Group I and 594.3 ± 297.87 cc in Group II). Liver dysfunction was also more pronounced in patients of ALA with ALD, as reflected in higher derangements of liver enzyme levels, bilirubin levels (both direct and total) and synthetic functions, bothat the time of admission, as well as discharge. (Table 2,3)
Treatments
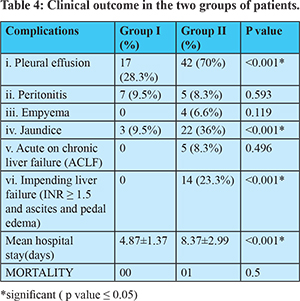
In this study four treatment options were employed: 1) Conservative management; 2) Needle Aspiration; 3) Pigtail Catheter insertion, and 4) Open surgery. In group I, 26(43.3%) patients were managed conservatively, 15 (25%) patients underwent needle aspiration, in 13 (21.6%) patientspigtail catheter was inserted, and 6 (10%) were managed with open surgery. In Group II only 3 (10.5%) patients were managed conservatively, 25 (41.6%) patients underwent needle aspiration, 27 (45%) patients had pigtail catheter insertion, and 5 (18.3%) were managed with open surgery. The indication for open surgery was rupture of abscess in the peritoneal cavity leading to peritonitis. (Table 4). As regards antibiotics, in group I, 2 (3.3%) patients had to be put on higher antibiotics because they were not responding to treatment and in group II, 23 (38.33%) patients needed to be started on higher antibiotics. 5(8.3%) patients in group II who developed acute decompensation were given human albumin.
Complications and Outcome
The most common complication in both groups was right-sided pleural effusion. The rate of complications in group II was significantly higher than in group I. (right pleural effusion 28% in Group I vs 70% in Group II; jaundice- 9.5% in group I vs 36 % in group II). Complications like empyema (6.6%), ACLF (8.3%) and impending liver failure (23.3%) were seen only in patients with ALD. Patients of ALA with ALD also had significantly longer periods of stay in the hospital (Group I was 4.87 ± 1.37 days and in Group II 8.37 ± 2.99 days). (Table 5)
Discussion
Alcohol intake has been identified as an important predisposing factor for ALA and is proposed to act through a multitude of mechanisms, including hepatic damage by alcohol, lowered body resistance and suppression of liver function due to poor nutritional status of habitual consumers of alcohol, increased presence of amoebae in the liquor prepared locally with poor regard to aseptic techniques, and depression of immune mechanisms in chronic alcoholics3. A study conducted in India showed that 67.5% of patients with amoebic liver abscesses were from the low socioeconomic class and 72% were alcoholics1. In our study, it was found that 76.6% of patients consumed alcohol. Further patients with ALD had a higher frequency, greater volume and longer duration of alcohol intake. Mean years of alcohol intake in patients with ALD was 8.39 years as compared to 2.92 years in those without ALD.
Alcoholics are also reported to have larger abscesses, a greater frequency of complications, and delayed resolution of the abscesses3. The results of our study are in concurrence with these findings. Group II patients had a larger size of abscesses in comparison to Group I (544 ± 297 cc and 341 ± 208 cc respectively). Right pleural effusion was the most common complication, followed byjaundice. Jaundice is a frequent occurrence in ALA, reported to be present in 6% to 29% of patients10. Many studies have shown that larger size of liver abscess is associated with jaundice.10,11 It is postulated that the main cause of jaundice in ALA is pressure and distortion of the biliary tree resulting in intrahepatic cholestasis10.
A higher rate of liver dysfunction was noted in patients of ALA with ALD. The mean SGOT, SGPT and ALP values both at admission and at the time of discharge were higher in the group with ALD than those without ALD. These results are in concurrence with previous studies showing deranged values1,3. The mean total bilirubin at the time of admission in group I was 1.2 mg/dl and 3.01 mg /dl in group II, while at discharge it was 1.1 mg/dl and 2.06 mg/dl in the two groups respectively. This small range of deranged value of serum bilirubin has been reported in earlier studies as well13. On comparing the synthetic function of the liver, patients with ALD had higher INR and lower albumins levels. Since the ALD patients have a poor reserve before infection, it results in a greater fall in the synthetic activity and greater mean bilirubin values.
In our study, a much smaller number of patients with ALD could be managed conservatively (10% as compared to 43% in group I). Further 38% of patients had to be started or shifted to higher antibiotics as they either did not show an adequate response or showed signs of impending failure. Intervention in the form of needle aspiration and PCD for ALA was required more frequently in the ALD group. The mean hospital stay recorded in group I and group II of this study were 4.87 days and 8.27 days respectively. Previous studies have also reported mean in-hospital stay of patients for 8-15 days1,12. There was one mortality seen in our study and the patient belonged to the ALD group. Other studies too report mortality in the range from 2-15%.12,13
Five (8.33%) patients in group II developed acute on chronic liver failure. Twenty-three per cent of patients were categorized as impending failure as they had coagulopathy ( INR = 1.5) and hypoalbuminemia and developed ascites and pedal oedema. These patients required more intensive care via drainage of the pus, higher antibiotics and correction of coagulopathy and hypoalbuminemia by fresh frozen plasma. In our patients, we administeredhuman albumin whenever they developed oedema or ascites and found that these infusions helped in the resolution of symptoms faster, while antibiotics and drainage of pus controlled infection.In literature too it is reported that systemic inflammation is a major issue in patients with decompensated cirrhosis and that albumin is a potent modulator of the innate immune system that could be useful in the management of ACLF14.
This is probably the first study that has objectively gathered evidence about the course of ALA in the presence of ALD and advocates the need for defining treatment protocol.
The limitation of our study is the non-probability sample size, which mandates the collection of larger data set in further studies to validate our observations.
Conclusion
In view of the more aggressive course of ALA in patients with ALD, it can be recommended that these patients should be started on higher antibiotics, have a lower threshold of abscess drainage and get actively managed for reversal of impending failure by replacement of albumin and clotting factors (FFP). A detailed assessment of ALD should be an important part of the workup of patients with ALA.
References
- Ghosh S, Sharma S, Gadpayle AK, Gupta HK, Mahajan RK, Sahoo R, et al. Clinical, laboratory, and management profile in patients of liver abscess from northern India. J Trop Med. 2014;2014:142382.
- Greenstein AJ, Barth J, Dicker A, Bottone EJ, Aufses AHJ. Amebic liver abscess: a study of 11 cases compared with a series of 38 patients with pyogenic liver abscess. Am J Gastroenterol. 1985 Jun;80(6):472–8.
- Mukhopadhyay M, Kumar A, Amitava S, Swadhin S. Amoebic liver abscess?: presentation and complications. 2010;(February):37–41.
- Osna NA, Affairs V, Health NI, Donohue TM, Affairs V, Health NI, et al. Alcoholic Liver Disease?: Pathogenesis and Current Management. 1996;1:147–61.
- Dugum M, Mccullough A. Review Article Diagnosis and Management of Alcoholic Liver Disease. 2015;3:109–16.
- Hong YS, Sinn DH, Gwak G, Cho J, Kang D, Paik Y. Characteristics and outcomes of chronic liver disease patients with acute deteriorated liver function by severity of underlying liver disease. 2016;22(14):3785–92.
- Debnath CR, Debnath MR, Khalid MS, Mahmuduzzaman M. Clinical profile of 250 cases of amoebic liver abscess. Mymensingh Med J. 2013;22(4):712–5.
- O’Brien CP. The CAGE questionnaire for detection of alcoholism: A remarkably useful but simple tool. JAMA - J Am Med Assoc. 2008;300(17):2054–6.
- Liumbruno GM, Bennardello F, Lattanzio A, Piccoli P, Rossetti G. Recommendations for the use of albumin and immunoglobulins. Blood Transfus. 2009;7(3):216–34.
- Nigam P, Gupta AK, Goyal BM, Joshi LD. Cholestasis in amoebic liver abscess. 1985;140–5.
- Sharma MP, Sarin SK. Amoebic liver abscess in a North Indian hospital--current trends. Br J Clin Pract. 1987 Jun;41(6):789–93.
- Sharma N, Sharma A, Varma S, Lal A, Singh V. Amoebic liver abscess in the medical emergency of a North Indian hospital. BMC Res Notes. 2010;3:21–4.
- Stanley SL. Amoebiasis. Lancet. 2003;361(9362):1025–34.
- Arroyo V, Clària J. Acute-on-Chronic Liver Failure, Human Serum Albumin, and Immune Modulation: The Beginning of an Exciting Adventure. Clin Gastroenterol Hepatol. 2018;16(5):633–6.