48uep6bbphidcol2|ID
48uep6bbphidvals|2944
48uep6bbph|2000F98CTab_Articles|Fulltext
Bisphosphonates are pyrophosphate analogues that bind at the bone mineral surface, where they potently inhibit osteoclast-mediated bone resorption. They are used in the treatment of hypercalcemia due to malignancy, osteolytic lesions from multiple myeloma, bone metastasis from solid tumours, osteoporosis, and Paget’s disease. They have also been used effectively in the medical management of primary hyperparathyroidism to decrease serum calcium.1,2 Inhibition of osteoclast activity in bone by bisphosphonates can result in hypocalcaemia and hypophosphatemia. The majority of patients do not manifest hypocalcaemia due to compensatory mechanisms such as raised Parathyroid Hormone (PTH). However, in patients with low Vitamin D levels, renal failure, prior parathyroidectomy, hypomagnesemia, and hypoparathyroidism, these compensatory mechanisms may be blocked, resulting in hypocalcaemia.3-5 Here we report a case in which the use of bisphosphonate for severe hypercalcemia due to primary hyperparathyroidism, led to life-threatening hypocalcaemia.
Case Report
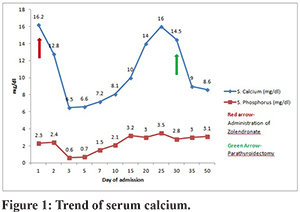
A 49-year-old female patient with a history of gall stones (post cholecystectomy) presented with complaints of severe upper abdominal pain with radiation to the back and recurrent vomiting for the past few hours. On examination, she was dehydrated with BP-120/80 mmHg, pulse-100/min, with abdomen distention, epigastric tenderness, and sluggish bowel sounds. Laboratory investigations revealed raised amylase (1545 U/l) and Lipase(3070 U/l), Leukocyte count-15000/mm3, hemoglobin-14.0g%, PCV-43, normal renal and liver function tests. USG Abdomen revealed bulky pancreas. Diagnosis of acute pancreatitis was made, and the patient was started on RT aspiration, IV fluids, and IV painkillers. Workup for aetiology revealed severe hypercalcemia (serum calcium-16.2 mg/dl), Albumin-4.6 mg/dl, and normal lipid profile. CRP-103 mg/L, high Parathyroid Hormone (PTH)-596pg/ml (Normal 12-88 pg./ml), low 25-OH Vitamin D levels-14ng/ml (Normal 30-100ng/ml), low Phosphorus 2.5 mg/dl (normal 2.7-4.5mg/dl), normal ACE levels and normal thyroid profile. Primary hyperparathyroidism was suspected. Aggressive fluid resuscitation was continued, and Inj Zoledronate 4 mg IV over 15 mins was given because of the hypercalcaemic crisis. She was shifted to ICU and required IV Fentanyl for control of pain. On Day 2, serum calcium decreased to 12.8 meq/l. Bedside Ultrasound neck was normal. On Day 3, the patient had seizures followed by bradycardia and cardiac arrest. She was revived after two cycles of CPR and required mechanical ventilation. Blood chemistry revealed severe hypocalcaemia(S. calcium-6.5 mg/dl) with low phosphorus-1.2 mg/dl and low magnesium-1.7 mg/dL (Normal 1.8-3.6mg/dL). Patient also had severe metabolic acidosis (pH-7.1,pco2-39,HCO3-14,lactate-5.6). Ionized Calcium was low-0.58 mmol/L (normal 1.1-1.3 mmol/L). She was started on injectable calcium gluconate(3g/day) and oral Vitamin D by Ryle’s tube. Contrast CT abdomen was suggestive of acute necrotizing pancreatitis with an acute peripancreatic fluid collection. Serum calcium improved gradually (Figure 1). IV calcium gluconate was given for eight days, followed by oral calcium (3g / day) with Calcitriol 0.25 mg twice a day and Vitamin D3 60k weekly. On Day 21, calcium started rising again, which was managed with hydration, saline diuresis, Inj Calcitonin (200 U s/c twice a day), and Tab Cinacalcet 30 mg twice a day. Haemodialysis had to be done as serum calcium remained high. Parathyroid MIBI scan showed Right inferior parathyroid adenoma. On Day 32 of admission, Right inferior parathyroidectomy was done. Calcium levels gradually decreased, and she was started on oral calcium supplementation (3g/day). Repeat CT abdomen suggestive of Walled off Necrosis. She was discharged on Day 50 of admission on oral calcium, magnesium, and Vitamin D3 supplements. Histology of surgical specimen confirmed parathyroid adenoma.
Discussion
Bisphosphonates are potent inhibitors of osteoclastic bone resorption. Hence serum calcium and phosphorus decrease, and intact parathyroid hormone (PTH) increases after its administration. Increased PTH increases the reabsorption of calcium in the kidneys and intestine. It also stimulates the kidneys to produce 1, 25-dihydroxyvitamin D leading to the preservation of calcium. Thus, bisphosphonate-induced hypocalcaemia often abates despite ongoing bisphosphonate therapy. Intravenous bisphosphonates (pamidronate, ibandronate, and zoledronic acid), which are more potent than oral bisphosphonates, may cause symptomatic hypocalcaemia mostly within days after infusion. Conditions that may impair compensatory increase in PTH may make patients prone to hypocalcaemia. Various studies have found that vitamin D deficiency, pre-existing hypoparathyroidism, renal failure, hypomagnesemia, and parathyroid dysfunction during thyroidectomy predispose patients to severe bisphosphonate-induced hypocalcaemia.3-5
Various studies have investigated the incidence of hypocalcaemia with zoledronic acid. In 120 patients who received zoledronic acid for malignancy, hypocalcaemia developed in 35 percent of patients. 8% of patients had symptomatic hypocalcaemia requiring IV calcium supplementation.5
The hypercalcaemic crisis is characterized by a serum calcium level greater than 14 mg/dL with severe signs of hypercalcemia that are reversible with the correction of hypercalcemia. Clinical findings include volume depletion, metabolic encephalopathy, gastrointestinal symptoms, renal and cardiovascular manifestations. Traditionally, the recommended treatment for hypercalcaemic crisis due to hyperparathyroidism is emergency parathyroidectomy within 72 hours. However, it was reported that there is a risk for cardiac arrhythmias when severe hypercalcemia is not treated preoperatively.
In primary hyperparathyroidism, bisphosphonates have been used for improving BMD in asymptomatic patients and in hypercalcaemic crisis for decreasing serum calcium. In a retrospective study of 177 patients with primary hyperparathyroidism, 37(21%) patients presented with a hypercalcaemic crisis, with 13.5% having pancreatitis. The use of pamidronate in 7 crisis patients resulted in a quicker lowering of serum calcium, and the incidence of postoperative hypocalcaemia was not high.1 Bisphosphonates have been used successfully as a bridge to parathyroidectomy in 7 patients with hyperparathyroid crisis in a study.2 However, there is no guideline for bisphosphonate use in hyperparathyroid crisis, and use is based on clinical experience only.
In our patient, PTH was significantly raised already, but serum 1,25-dihydroxyvitamin D levels and magnesium levels were low. Zoledronate was used as an emergency measure to decrease toxic serum calcium levels. Severe hypocalcaemia may have been due to impaired compensatory mechanisms and protracted action of zoledronate. Another peculiar observation is that hypocalcaemia occurred within 2-3 days of Zoledronate injection as it has been described to occur later in previous studies.
Bisphosphonates have gained popularity in the treatment of hypercalcemia due to good efficacy and prolonged action. However, evaluation of Vitamin D deficiency, renal function, PTH levels, and serum magnesium should be performed before administration. Correction of Vitamin D and magnesium levels should be done before the injection of bisphosphonate.
References
- Singh DN, Gupta SK, Kumari N, et al. Primary hyperparathyroidism presenting as hypercalcaemic crisis: Twenty-year experience. Indian Journal of Endocrinology and Metabolism.2015;19(1):100-105.
- Phitayakorn R, McHenry CR. Hyperparathyroid crisis: Use of bisphosphonates as a bridge to parathyroidectomy. J Am Coll Surg. 2008;206:1106–15.
- Peter R, Mishra V, Fraser WD. Severe hypocalcaemia after being given intravenous bisphosphonate. BMJ. 2004 Feb 7;328(7435):335-6.
- Maalouf NM, Heller HJ, Odvina CV, Kim PJ, Sakhaee K. Bisphosphonate-induced hypocalcaemia: report of 3 cases and review of literature. EndocrPract. 2006 Jan-Feb;12(1):48-53.
- Chennuru S, Koduri J, Baumann MA. Risk factors for symptomatic hypocalcaemia complicating treatment with zoledronic acid. Intern Med J. 2008 Aug;38(8):635-7.