48uep6bbphidcol2|ID
48uep6bbphidvals|1889
48uep6bbph|2000F98CTab_Articles|Fulltext
Hepatocellular carcinoma (HCC) constitutes more than 90% of primary liver cancers and is a major global health problem1. Peritoneal dissemination of hepatocellular carcinoma (HCC) is a rare presentation, with an incidence of 2% to 6% detected during autopsy or laparoscopy2. Gastrointestinal tract involvement is noted in 4-10% of cases and mostly via direct invasion and hematogenous metastasis and is rather rare3. To the best of our knowledge, HCC with peritoneal metastasis diagnosed by trans rectal endoscopic ultrasound guided biopsy has not been reported previously. The current report is of 2 cases of metastatic HCC one each to peritoneum and stomach presenting with abdominal mass and gastrointestinal bleeding.
Case Report
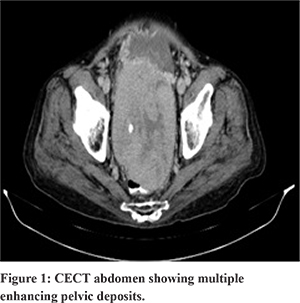
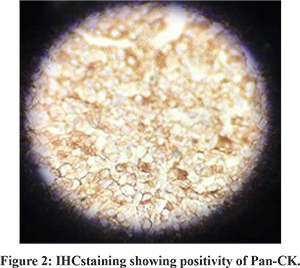
Patient A: A 63 year old male diagnosed case of decompensated chronic liver disease presented with abdominal pain and significant weight loss of 10 kgs for 1 month duration. On examination, he had palpable liver without any bruit, ascites and an ill-defined pelvic mass. Ultrasound abdomen followed by contrast enhanced computed tomography (CECT) abdomen revealed multiple lesions in both lobes of liver enhancing in the arterial phase- largest measuring 2 X 1.8 cm. There were multiple enhancing pelvic deposits- largest measuring 15 X17 cm and moderate ascites (Figure 1). He was non reactive for hepatitis B surface antigen and hepatitis C virus. Alpha fetoprotein (AFP) levels were elevated- 414 ng/mL (normal:<10ng/mL). Large pelvic deposit is unusual in HCC, so an endoscopic ultrasound (EUS) guided biopsy was done. Transrectal EUS revealed 7x5 cm heteroechoic pelvic mass with moderate ascites in pararectal area. Fine needle aspiration biopsy was done and cellular smears comprising of sheets, clusters and trabeculae of round to polygonal neoplastic cells with abundant clear cytoplasm were seen. Immunohistochemistry (IHC) revealed Hepatocyte Paraffin (HepPar1): Negative, AFP: Positive, Alfa 1 anti-trypsin: Negative ,Pan CK: Positive; Cytokeratin (CK) 7: Negative (Figure 2). Despite inconclusive Hep Par1 immunostain a diagnosis of Clear cell variant of hepatocellular carcinoma (CHCC) was made as all other clear cell tumors were ruled out by IHC and imaging. He was treated with sorafenib, but succumbed to illness after 4 months of diagnosis.
Patient B: A 77 year old male, diagnosed case of HCC BCLC stage D on sorafenib for 4 months, under regular follow up with primary endoscopic variceal ligation schedule presented with hematemesis and melena. Emergency endoscopy was performed which revealed small esophageal varices, diffusely hyperemic stomach with multiple submucosal lesions with surface ulceration and hemorrhage (Figure 3). Microscopic examination revealed round to polygonal cells with pleomorphic hyperchromatic nuclei, prominent nucleoli and eosinophilic cytoplasm suggestive of malignancy. The results of IHC stains were positive for Hep Par 1 and negative for CK7, CDX-2. These histologic features resulted in the diagnosis of metastatic HCC.
Discussion
HCC is the fourth and eighth leading cause of cancer related mortality in “India specific” men and women respectively4. The frequency of peritoneal seeding of HCC is noted to occur in 5.6%-14.5% following spontaneous rupture of HCC and tumor seeding5. Our case had peritoneal metastasis without any clinical evidence of tumor rupture. The cause of spread might be hematogenous. Cytoreductive surgery for HCC with peritoneal metastasis is related to severity of CLD, tumor involvement and response to surgery determines the prognosis of advanced HCC. In our first case with CTP C status, the extrahepatic metastases to peritoneum precluded any possibilities of surgery, transplantation and any radiological interventional procedure (balloon occluded transarterial chemoembolisation).
Gastrointestinal involvement in HCC is reported in 0.5-2% of all HCC cases. Hypervascular exophytic subcapsular large > 5 cm HCC with or without transarterialchemoembolisation (TACE) can directly invade stomach, duodenum, jejunum or colon. Only about 30 cases of direct invasion of HCC to stomach are reported in English literature so far6.
References
- LlovetJosep M., DucreuxM, European Association For The Study Of The Liver, European Organisation for Research and Treatment of Cancer EASL–EORTC clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 2012;56:908–943.
- Yeh CN, Chen HM, Chen MF, Chao TC. Peritoneal implanted hepatocellular carcinoma with rupture after TACE presented as acute appendicitis. Hepatogastroenterology. 2002;49:938-40.
- Hu ML Tai WC, Chuah SK, Chiu YC, Wu KL, et al. Gastric metastasis of hepatocellular carcinoma via a possible existing retrograde hematogenous pathway. J Gastroenterol Hepatol. 2010; 25: 408-412.
- Kumar A, Acharya SK, Singh SP , et al. The Indian National Association for Study of the Liver (INASL) Consensus on Prevention, Diagnosis and Management of Hepatocellular Carcinoma in India: The Puri Recommendations. J Clin Exp Hepatol. 2014; 4:S3-26.
- Chauhan U, Rajesh S, Kasana V, Gupta S, Bihari C. Spermatic Cord and Peritoneal Metastases from Unruptured Hepatocellular Carcinoma. JCDR. 2015;9(10):TD04-TD05.
- Korkolis DP, Aggeli C, Plataniotis GD, et al. Successful en bloc resection of primary hepatocellular carcinoma directly invading the stomach and pancreas. World Journal of Gastroenterology?: WJG. 2009;15(9):1134-1137.