48uep6bbphidcol2|ID
48uep6bbphidvals|1839
48uep6bbph|2000F98CTab_Articles|Fulltext
Idiopathic pulmonary hemosiderosis (IPH) is manifest as a triad of pulmonary symptoms, alveolar opacities on chest radiographs, and iron deficiency anemia. The presence of hemosiderin in macrophages obtained in gastric or bronchoalveolar lavage is considered crucial in the diagnosis of pulmonary hemosiderosis. IPH in association with celiac disease (CD), known as Lane-Hamilton syndrome, could be due to the fact that both entities share a common pathogenic immune pathway. There are limited numbers of case reports of this syndrome in literature.
Case Report
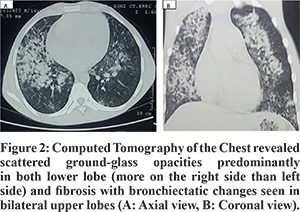
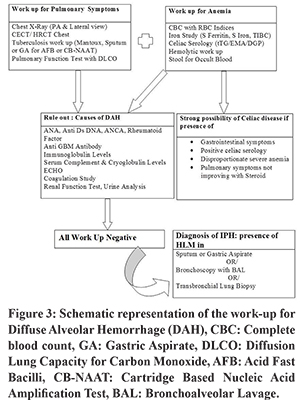
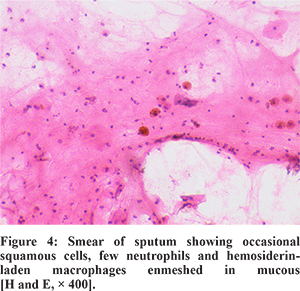
A 14 years old non-smoker adolescent boy presented with complaints of cough associated with intermittent hemoptysis and history of progressive pallor for last 5 months. Cough was present off and on with mild specks of blood. No other significant history was present. Past and family history were unremarkable. On admission, he had severe pallor, tachycardia (pulse rate 140/min) and tachypnea (respiratory rate 42/min) with normal blood pressure and growth parameters. On auscultation, bilateral infrascapular crackles and hyperdynamic precordium with a hemic murmur was present. There was no hepatosplenomegaly. Investigations revealed severe anemia (Hb of 2 gm/dl), ESR of 10 mm/1st hour; total leukocytecount of 10400/mm3 and platelet count of 3.36 lacs/mm3. He had been investigated for anemia and blood investigations demonstrated iron deficiency anemia (peripheral smear showed microcytic, hypochromic anemia, serum ferritin 5 ng/ml, serum iron 30 µg/dl and TIBC 393 µg/dl). Renal function tests, liver function tests, C-reactive protein, coagulation studies and urine analysis were within normal limits. Chest radiograph demonstrated bilateral lower zone alveolar type opacities (Figure 1A). A contrast-enhanced computed tomography scan of the chest revealed scattered ground-glass opacities predominantly in both lower lobe and fibrosis with bronchiectatic changes seen in bilateral upper lobes (Figure 2A and 2B). Work-up for pulmonary tuberculosis and HIV ELISA were negative. All laboratory tests for diffuse alveolar hemorrhage (DAH) were negative as mentioned in Figure 3. Cardiac evaluation was normal. Pulmonary function tests showed a restrictive pattern with FEV1 63%, FVC 68%, FEV1/FVC 95% and PEFR 78% of predicted. In sputum examination, the smear showed occasional squamous cells, few neutrophils and hemosiderin-laden macrophages (HLM) enmeshed in mucous, suggesting the possibility of intra-alveolar hemorrhage (Figure 4). The presence of bilateral ground-glass haziness and iron deficiency anemia along with HLM in sputum examination with an exclusion of other causes confirmed the diagnosis of IPH.
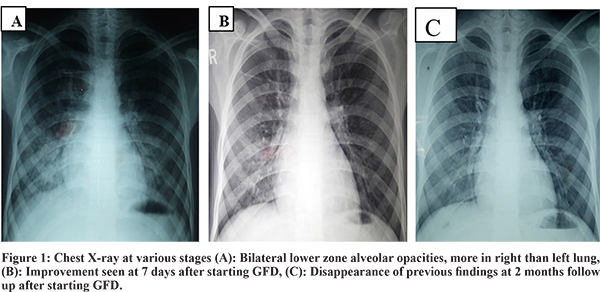
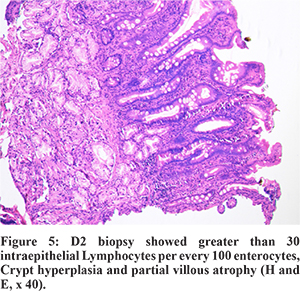
Though there were no gastrointestinal symptoms in our case, the possibility of CD was considered in view of severe iron deficiency anemia (degree of anemia was out of proportion to the chest imaging findings) and previously described association between IPH and CD. Positive serology (Anti-tissue transglutaminase (tTG) IgAtitres>100 AU/ml, normal <10 AU/ml), along with the presence of scalloping in second part of duodenum on Esophagogastroduodenoscopy (EGD) and modified Marsh grade 3A histopathology finding on duodenal biopsy (Figure 5) were consistent with a diagnosis of CD. Based on this, afinal diagnosis of Lane-Hamilton syndrome (CD with IPH) was made. Child was initially managed with blood transfusions and put on a gluten-free diet (GFD). Pulmonary symptoms completely resolved after few days. Hemoglobin at 2 months follow up was normal with complete disappearance of previous radiological findings in chest X-Ray (Figure 1B, 1C). Long-term follow up was advised to check the compliance of GFD and recurrence of sign or symptoms.
Discussion
IPH is a rare disease of unknown etiology, predominantly affects children and adolescent and incidence varies from 0.24 to 1.23 patients per million children.1 Cough, anemia and failure to thrive with or without hemoptysis is the usual presentation in children. Diagnosis of IPH requires evidence of DAH and exclusion of other causes of DAH.2 Diagnostic work-up for the evaluation of IPH/DAH is shown schematically in Figure 3. A diagnosis by lung biopsy is considered gold standard. But many authors have accepted the presence of HLM in sputum, gastric or broncho-alveolar lavage (BAL) fluid as diagnostic if typical clinical features are present and are not accompanied by evidence of extrapulmonary disease.In 1971, Lane and Hamilton described the association of IPH with CD in a young man for the first time.3 While immunological mechanisms are believed to be effective in the formation of IPH and CD, a pathogenic link between them cannot be precisely understood. Routine screening of CD is recommended in all cases of IPH, even in the absence of gastrointestinal symptoms by many authors.4,5
In Lane-Hamilton syndrome, a gluten-free diet for CD, with or without steroids for IPH, is the mainstay of therapy. The clinical importance of this association is that a significant improvement can be obtained with gluten-free diet not only in intestinal but also in pulmonary symptoms. Use of gluten-free diet (GFD) results in partial or complete cessation of lung hemorrhage and reduced the need for blood transfusions and steroid therapy in Lane-Hamilton syndrome.4,5 In a report of 20 patients with IPH associated with CD, GFD alone had been prescribed for 16 of them and pulmonary symptoms were improved in 12 patients.4 In Lane-Hamilton syndrome, gastrointestinal symptoms are reported to occur in about 50% of cases only.3 Emphasising this, our patient had no prior history of diarrhea, steatorrhea, flatulence or weight loss. Pulmonary symptoms improved on a gluten-free diet alone within 7 days of admission with complete resolution of chest X-ray findings 2 months after starting GFD.
In conclusion, a high index of suspicion for celiac disease should be kept in patients with pulmonary hemosiderosis, especially with disproportionately severe anemia inspite of absence of gastrointestinal symptoms and vice-versa. Non-invasive investigations such as sputum examination for hemosiderin-laden macrophages can be considered diagnostic. This entity is important to recognize as treatment with a gluten-free diet alone can lead to remission of the pulmonary symptoms.
References
- Ioachimescu OC, Sieber S, Kotch A. Idiopathic pulmonary haemosiderosis revisited. Eur Respir J 2004; 24: 162–70.
- Susarla SC, Fan LL. Diffuse alveolar hemorrhage syndromes in children. Curr Opin Pediatr 2007; 19: 314-320.
- Lane DJ, Hamilton WS. Idiopathic steatorrhoea and idiopathic pulmonary haemosiderosis. Br Med J. 1971; 2: 89–90.
- Agarwal R, Aggarwal AN, Gupta D. Lane-Hamilton syndrome: simultaneous occurrence of coeliac disease and idiopathic pulmonary haemosiderosis. Intern Med J. 2007; 37(1): 65-67.
- Sethi GR, Singhal KK, Puri AS, Mantan M. Benefit of gluten-free diet in idiopathic pulmonary hemosiderosis in association with celiac disease. Pediatr Pulmonol. 2011; 46: 302-305.