|
Tarun Kumar1, Maitrayee Roy1, Rajni Yadav1, Hemant Jain2, Sujoy Pal2, Prasenjit Das1, Siddhartha Datta Gupta1 1Department of Pathology, 2Department of Gastrointestinal Surgery, All India Institute of Medical Sciences, New Delhi, India.
Corresponding Author:
Dr Rajni Yadav Email: drrajniyadav@gmail.com
DOI:
http://dx.doi.org/10.7869/tg.428
48uep6bbphidcol2|ID 48uep6bbphidvals|1810 48uep6bbph|2000F98CTab_Articles|Fulltext Periampullary neoplasms collectively include carcinomasarising from the duodenum, ampulla of Vater, distal part of the common bile duct and head of pancreas.1 Ampullary carcinoma constitutes 7% of all peri-ampullary carcinomas with adenocarcinoma being the commonest histologic type.2 Osteoclast giant cells (OGCs) have been reported to be seen in various malignanciesarising from the pancreas, breast and rarely the liver, kidney, urinary bladder, gall bladder and the endometrium.3,4 Ampullary adenocarcinoma with OGCs is an extremely rare entity and has only sparsely been described in literature till date.5
Case Report
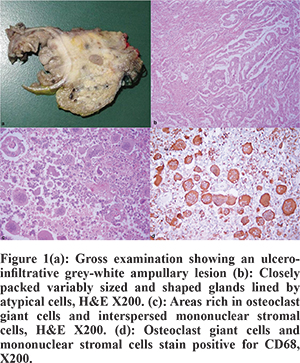
A 53 year-old post-menopausal female presented with yellowish discoloration of the eyes, clay-colored stools and high-colored urine for 6 months. She reported a loss of appetite, an unquantified loss of weight, occasional pain abdomen radiating to the back and multiple episodes of cholangitis. She had a history of endoscopic retrograde cholangio-pancreatography (ERCP) followed by laparoscopic cholecystectomy 5 months back at a private hospital. Subsequently, in view of the recurrent cholangitis, repeat ERCP and stent exchange was performed thrice before she was referred to our hospital.Computed tomography scan of the abdomen showeda mass in the peri-ampullary region causing dilatation of the common bile duct and the pancreatic duct along with atrophy of the body and tail of pancreas. An MRI showed a hyperintenseperi-ampullarylesion on T1-weighted images. A diagnosis of adenocarcinoma was made following an endoscopic ultrasound-guided biopsy. The patient subsequently underwent a pylorus-resectingpancreatico-duodenectomyalong with appropriate lymphadenectomy. A grey-white ulcero-infiltrative tumour measuring 4 cm x 3 cm x 1.8 cm was identified in the ampullary and periampullary region of the specimen (Figure 1). Microscopic evaluation revealed a tumour composed of variably sized and closely packed glands lined by tumour cells exhibiting nuclear enlargement with pleomorphism and at places, prominent nucleoli were seen. Mucin was also seen in the glandular lumina. The tumour showed areas rich in OGCs and interspersed mononuclear stromal cells. The adenocarcinoma areas were intimately admixed with OGC rich areas. Immunohistochemically, the tumor cells showed positivity for cytokeratin 7 (CK7) and were negative for CD68 while the OGCs and mononuclear stromal cells were positive for CD68 and negative for CK7. The tumor infiltrated the duodenal wall and superficially involved the adjacent pancreas. The non-involved pancreas showed features of chronic pancreatitis. Perineural and lympho-vascular invasion were seen. The dissected lymphnodes showed tumour metastases. However, the metastases was composed of the adenocarcinoma component only. A final diagnosis of moderately differentiated adenocarcinoma with osteoclast giant cells was offered. The pathological stage was T3N1M0, IIB. The postoperative course was uneventful except for mild superficial surgical site infection. However, the patient succumbed to death 4-5 months post-surgery.
Discussion
Ampullary carcinomas are rare tumorsthat account for approximately 0.5% of all gastrointestinal malignancies. Adenocarcinoma(65%) is the most common histologic type usually showing either an intestinal or pancreato-biliary type of differentiation. The less common histological subtypes include mucinous, signet ring cell, neuroendo-crine and undifferentiated carcinomas.6 Westgaard et al7 demonstrated that histological subtyping has prognostic implications with the intestinal subtype showing a better prognosis compared to the pancreaticobiliary type. Other prognostic factors include tumor size, grade, lymph node status, perineural invasion, vascular invasion and loco-regional infiltration.7 Giant cell tumours (GCTs) can be seen in a wide variety of sites including the pancreas, breast, thyroid, parotid, gall bladder, colon, skin, orbit, kidney, heart and soft tissue. In the pancreas, three kinds of GCTs are known-namely osteoclastic, pleomorphic and mixed types. Subtyping is considered important as the osteoclastic subtype has a better prognosis than the pleomorphic type.3 The exact mechanism of OGCs formation in tumours is not known. The histogenesisis controversial, with suggestions of both epithelial and mesenchymal origin. The pure osteoclastic subtype that resembles giant cell tumor of bone is probably mesenchymal in origin as it is composed of OGCs and mononuclear stromal cells only and lacks epithelial elements. The OGCs and stromal cells stain positive forvimentin and CD68 while are non-reactive for cytokeratin, thereby, confirming a mesenchymal origin. In contrast, the pleomorphic subtype, which does not reveal any particular morphologic epithelial differentiation, shows positivity for cytokeratin and negativity for CD68 and vimentin favouring an epithelial origin.3,8 However, there seems to be an overlap between the above discussed types as a mixed subtype also exists. The mixed subtype can have well defined carcinomatous areas as well as a stromal component rich in OGCs. Our case was a mixed one as it had areas of adenocarcinoma and other areas rich in OGCs. Shishido-Hare et al4 demonstrated in breast cancer that the appearance of OGCs in stroma is due to the pro-tumoral differentiation of macrophages as aresult of the hypervascular micro-environment induced by cancer rather than an antitumoral immunological reaction. Presence of OGCs in breast carcinomas has been suggested to impart a less favorable outcome.4 The origin and role of OGCs in various tumors is, thus, controversial and the behavior of these tumours is also widely variable. The presence of components other than OGCs such as glandular, pleomorphic and stromal does seem to greatly influence the behavior of giant cell rich tumors. Our case showed an aggressive behavior despite the absence of marked pleomorphism which is most likely due to the presence of the conventional glandular component, perineural invasion, lympho-vascular invasion and lymph nodal adenocarcinomatous metastatic deposits. Surgical resection is considered the first line of treatmentand the role of adjuvant therapy is unclear. Radiotherapy has been tried in view of the existing knowledge regardingthe radio-sensitivity of GCTs of the bone. Chemotherapeutic agents like gemcitabine may be used in cases of disseminated disease or incomplete resections.8 To conclude, ampullary adenocarcinoma with OGCs is a rare entity and shows an aggressive behavior, thus, suggesting the need for intensive multimodality management.
References - Kimura W, Futakawa N, Zhao B. Neoplastic diseases of the papilla of Vater. J Hepatobiliary Pancreat Surg. 2004;11:223-31.
- O’Connell JB, Maggard MA, Manunga J, Tomlinson JS, Reber HA, Ko CY et al. Survival after resection of ampullary carcinoma: a national population-based study. Ann Surg Oncol. 2008;15:1820-7.
- Temesgen WM, Wachtel M, Dissanaike S. Osteoclasticgiant cell tumor of the pancreas. Int J Surg Case Rep. 2014;5:175-9.
- Shishido-Hara Y, Kurata A, Fujiwara M, Itoh H, Imoto S, Kamma H. Two cases of breast carcinoma with osteoclastic giant cells: Are the osteoclastic giant cells pro-tumoural differentiation of macrophages? Diagn Pathol. 2010;5:55.
- Matsuzawa G, Shirabe K, Gion T, Tsujita E, Ooya M, Kajiyama K. Surgically resected undifferentiated carcinoma with osteoclast-like giant cells of the periampullary region involving the orifice of the papilla of Vater: Report of a case. Surg Today. 2010;40:376-9.
- Fischer HP, Zhou H. Pathogenesis of carcinoma of the papilla of Vater. J Hepatobiliary Pancreat Surg. 2004;11:301-9.
- Westgaard A, Tafjord S, Farstad IN, Cvancarova M, Eide TJ, Mathison O et al. Pancreatobiliary versus intestinal histologic type of differentiation is an independent prognostic factor in resected periampullary adenocarcinoma. BMC Cancer. 2008;8:170-4.
- Moore JC, Bentz JS, Hilden K, Adler DG. Osteoclastic and pleomorphic giant cell tumors of the pancreas: a review of clinical, endoscopic, and pathologic features. World J Gastrointest Endosc. 2010;2:15-9.
|