|
|
|
|
 |
 |
| |
 |
|
|
Case Report |
|
|
|
|
|
Keywords :
|
|
|
P Naranje1, U Gorsi1, L Gupta1, A Bhalla2, N Khandelwal1
1Department of 1Radiodiagnosis and Imaging and 2Medicine Post Graduate Institute of Medical Education and Research, Chandigarh, India
Corresponding Author:
Priyanka Naranje
E mail: priyanka11sh@gmail.com
DOI:
http://dx.doi.org/10.7869/tg.327
48uep6bbphidcol2|ID 48uep6bbphidvals|1708 48uep6bbph|2000F98CTab_Articles|Fulltext Aortoenteric fistulas (AEF) are communications between the aorta and a loop of the bowel. Primary AEF is a rare and challenging entity and requires awareness and a vigilant approach to reach the diagnosis. Autopsy findings suggest that the incidence of primary AEF range between 0.04% and 0.07%.[1] Despite advances in technology, an AEF is difficult to diagnose and treat and is associated with high mortality (65-100%) and morbidity.[2] The radiologist plays a pivotal role in reaching an accurate and precise diagnosis of AEF. Imaging tools such as ultrasound and multidetector contrast enhanced computed tomography (CECT) helps in making an accurate diagnosis and to differentiate it from other inflammatory mimickers. We describe a case report of a 46 year old woman presenting to the emergency with shock and a history of melena and was subsequently accurately diagnosed to have an aortoenteric fistula. The objective of presenting this case report is to discuss the radiological findings of primary aortoenteric fistula in a patient presenting with acute hemodynamic instability.
Case Report
A 46 year old woman was referred to the emergency with melena and shock. She had a history of sudden abdominal pain, vomiting and weakness for one day. She had a past history of on and off fever for the last 1 year with loss of weight and appetite. There was no history of previous abdominal vascular surgery. On systemic examination, the patient was found to have pallor, feeble pulse and was hemodynamically unstable with a blood pressure (BP) of 80/50, heart rate (HR) of 138 beats per minute, C reactive protein (CRP) of 96 and hemoglobin (Hb) of 6.2gm%. The patient had a further drop in hemoglobin to a Hb of 3.7gm% within 12 hours. To search for the cause of the sudden drop in hemoglobin and patient’s worsening condition, a multidetector contrast enhanced computed tomography (CECT) was acquired on a 64 slice scanner (GE healthcare) by giving 100ml of iv contrast (300 mg /l) at the rate of 4.0 ml/sec and arterial phase images were acquired by bolus tracking followed by portal venous and delayed phase at 65 and 180 seconds respectively. However, despite being on resuscitative fluids and triple inotropes, the patient progressed to become severely hypotensive and had a cardio respiratory arrest from which she could not be revived even after cardiopulmonary resuscitation. The relatives of the patient didn’t give consent for the autopsy.
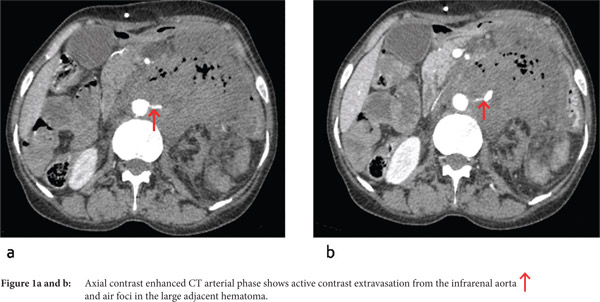
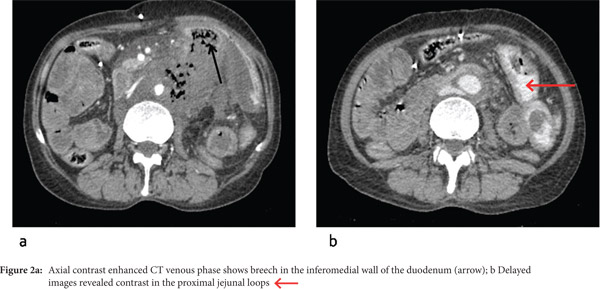
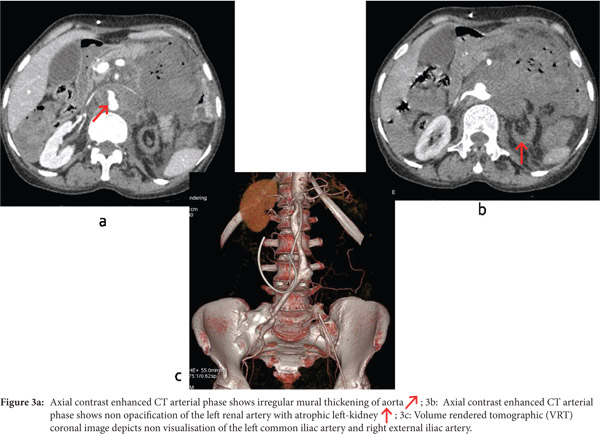
Multidetector CECT revealed the presence of an active contrast extravasation from the infrarenal aorta in the left paraaortic location forming a large hematoma measuring 8.2 x 7 x 9.2 cm. Multiple air foci were seen adjacent to the aorta and in the hematoma, with direct communication with the fourth part (D4) of the duodenum (Figure 1a,b). A breech of 2.4 cm was seen in the posterior wall of the D4 segment of the duodenum (Figure 2a). The delayed images revealed the presence of contrast in the proximal jejunal loops (Figure 2b). The abdominal aorta, from the level of origin of the superior mesenteric artery to its bifurcation shows circumferential asymmetric mural thickening with irregular luminal narrowing (Figure 3a). Additional findings included chronic thrombosis of the left renal artery with atrophic left kidney and chronic thrombosis of the left common iliac artery and right external iliac artery (Figure 3b,c).
Discussion
An aortoenteric fistula (AEF) is the abnormal communication between the aorta and the bowel loop. It can be primary or secondary, based on the history of abdominal aortic aneurysm (AAA) repair and the presence of prosthetic materials. Primary AEFs are rare and challenging entities and require awareness and a vigilant approach for its diagnosis. Most primary AEFs are seen in the duodenum (60%), arising in the 3rd and 4th part of the duodenum due to close proximity and direct contact with the abdominal aorta. The other 40% are associated with other loops of small and large bowel such as jejunum (12%), ileum (18%), cecum (8%), and appendix (4%).[3] Approximately 99% of primary AEFs have been estimated to arise from an abdominal aortic aneurysm, mostly atherosclerotic. Remaining cases were caused by radiation, pancreatic carcinoma, ulcers, diverticulitis, appendicitis and metastases.[4] The most common cause is a ruptured atherosclerotic abdominal aortic aneurysm that occurs secondarily to the pressure of the pulsating aneurysm on the adjacent bowel loop wall resulting in pressure necrosis.
Among the wide armamentarium offered by radiology, the multidetector contrast enhanced helical CT scan is the first line and the most preferred imaging modality of choice with sensitivity of 40-90% and specificity of 33-100%.[5] Other studies such as upper GI endoscopy, angiography and nuclear studies such as scintigraphy with SPECT using radiotracers gallium 67 citrate, 111 In labeled white blood cells or 99mTc hexametazime, can be helpful. However CT is by far the best modality owing to its widespread availability, short acquisition time and high resolution. The direct signs are the presence of ectopic gas around and within the aorta and the intravenous contrast in the gastrointestinal tract. Indirect signs include adjacent bowel wall thickening, disruption of the aortic fat cover and aortic wall, tethering of bowel loop to the anterior wall of the aorta and retroperitoneal hematoma or hematoma within bowel wall or lumen. Based on the above described signs, the diagnosis of a primary aortoenteric fistula was made in our patient. The list of differential diagnosis includes retroperitoneal fibrosis, infected aortic aneurysm, infectious aortitis, perigraft infection without fistulisation. The incidence of mortality in the untreated primary AEF is almost 100%, thus it warrants urgent treatment. The survival after surgery ranges from 18% to 93%, depending on the hemodynamic instability of the patient and experience of the surgeon.[6] Thus the radiologist plays a pivotal role in making an early diagnosis of this lethal entity.
References
- Voorhoeve R, Moll FL, de Letter JA, Bast TJ, Wester JP, Slee PH. Primary aortoenteric fistula: report of eight new cases and review of the literature. Ann Vasc Surg. 1996;10:40-8.
- Hughes FM, Kavanagh D, Barry M, Owens A, MacErlaine DP, Malone DE. Aortoenteric fistula: a diagnostic dilemma. Abdom Imaging. 2007;32:398-402.
- Champion MC, Sullivan SN, Coles JC, Goldbach M, Watson WC. Aortoenteric fistula. Incidence, presentation recognition, and management. Ann Surg. 1982;195:314-7.
- Gad A. Aortoduodenal fistula revisited. Scand J Gastroenterol Suppl. 1989;167:97-100.
- Vu QD, Menias CO, Bhalla S, Peterson C, Wang LL, Balfe DM. Aortoenteric fistulas: CT features and potential mimics. Radiographics. 2009;29:197-209.
- Kavanagh DO, Dowdall JF, Younis F, Sheehan S, Mehigan D, Barry MC. Aorto-enteric fistula: changing management strategies. Ir J Med Sci. 2006;175:40-4.
|
|
|
 |
|
|