|
Avishek Bagchi1, Kaustubh Mahamine1, Samiran Nundy2, Paras Kathuria1, Pabitra Sahu1, Suresh Kumar1, Naresh Kumar1, Premashis Kari
Department of Medicine1,
Maulana Azad Medical College,
Sir Ganga Ram Hospital2,
New Delhi, India
Corresponding Author:
Dr. P. Kar
Email: premashishkar@gmail.com
DOI:
http://dx.doi.org/10.7869/tg.308
48uep6bbphidvals|1372 48uep6bbphidcol2|ID 48uep6bbph|2000F98CTab_Articles|Fulltext GIST is the most common mesenchymal neoplasm of the gastrointestinal (GI) tract representing 0.1-3% of all gastrointestinal malignancies. GIST cells are thought to arise from a common precursor cell which also gives rise to intestinal cells of Cajal. GIST results from activating mutations in receptor protein kinase: either KIT (CD 117) or PDGFRA (platelet derived growth factor receptor alpha). The stomach and intestine are common sites of GIST. The periampullary region is a very rare site for GIST. To the best of our knowledge, only three cases of periampullary GIST have been reported before.[1,2,3]
Methods
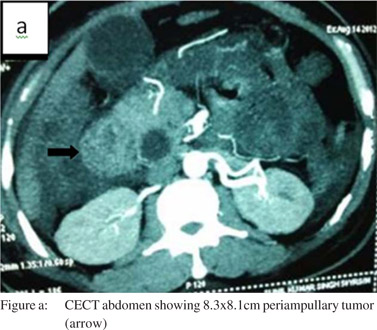
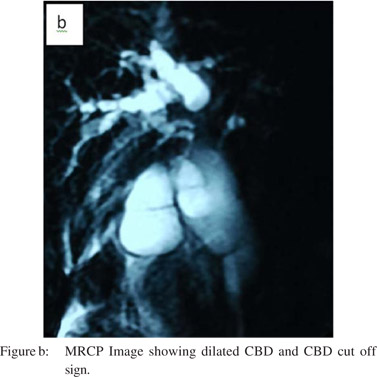
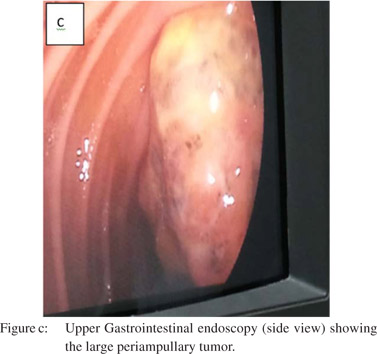
A 48-year-old man presented with progressive jaundice, anorexia and weight loss for 2 months, fever and abdominal pain for 7 days. On examination the patient was pale and icteric. There was firm, tender hepatomegaly 5 cm below the subcostal margin. Investigations showed microcytic hypochromic anemia (hemoglobin-6.3g%) with leucocyte count 12900cells/mm3 and neutrophils 85%. The stool was positive for occult blood. The patient had conjugated hyperbilirubinemia with total and direct bilirubin of 4.6 mg/dL and 4.0 mg/dL, respectively and alkaline phosphatase of 693U/L. Ultrasonography of the abdomen showed liver size 16 cm with dilatation of IHBR and common bile duct and 8.8×4.8×5.5 cm heterogenous hyperechoic mass near the head of pancreas in the periampullary region. CECT abdomen revealed 8.3 cm x8.1 cm well defined lesion in the region of the head of pancreas (Figure a). MRCP showed a well-defined lobulated hyperintense lesion in the region of head and uncinate process of pancreas with sudden cutoff of common bile duct (Figure b) and pancreatic duct at the lesion, which was suggestive of malignancy. Upper gastrointestinal endoscopy (Figure c) was done and multiple biopsies were taken from the mass. Endoscopic retrograde cholangiopancreatography failed to relieve biliary obstruction because of the large mass at the ampulla.
Bile mixed with pus was drained on percutaneous transhepatic biliary drainage and its culture was positive for E. coli. The patient was treated with antibiotics as per the sensitivity report. Subsequently Whipple’s pancreaticoduodenectomy (single loop anastomosis) with feeding jejunostomy was performed.
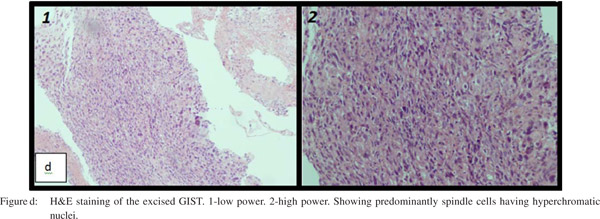
Histopathology of operated specimen (Figure d) revealed a malignant gastrointestinal stromal tumor (GIST) with high mitotic activity (>10/50hpf) with neural differentiation. Immunohistochemical studies were positive for S-100 and focally positive for CD1, negative for CD34, SMA and c-kit. Patient was started on Imatinib and is stable after 7 months of follow up.
Discussion
The most common site for GIST (gastrointestinal stromal tumor) is the stomach (50-60%) followed by small bowel (20-30%), large bowel (10%), esophagus (5%) and elsewhere in the abdomen 5%. Only 4% of GISTs occur in the duodenum and very rarely in the periampullary region. Mean age of presentation of this tumor is 60 years (40-80 years) with no predilection for either gender. Familial GIST are associated with Neurofibromatosis type I and Carney triad (GIST, paraganglioma, pulmonary chondroma).
Diagnosis of gastrointestinal stromal tumor depends on immuno-histochemical studies. Histologically GIST are classified as neural type, epitheloid type and mixed type.[4] Biological markers used in its diagnosis are c-kit (CD117) with positivity in>95% cases, CD34 positive in 60-70%, PDGFRA (platelet derived growth factor alpha) positive in<5% cases, vimentin and smooth muscle actin positive in 15-60%. Others likedesmin, S-100, keratin are rare. Treatment of primary GIST is complete surgical resection. Tumor designated as high risk should be treated with adjuvant Imatinib (tyrosine kinase inhibitor). Imatinib is the mainstay of treatment for metastatic or unresectable tumor. Tumor resistant to Imatinib is treated with Sunitinib.
References
- Filippou DK, Pashalidis N, Skandalakis P, Rizos S. Malignant gastrointestinal stromal tumor of the ampulla of Vater presentingwith obstructive jaundice. J Postgrad Med. 2006;52:204–6. (PMID: 16855323)
- Millonig G, Giesel EL, Reimann EM, Topalidis T, Seitz HK, Meuller S. Gastrointestinal stromal tumor in a patient with undiagnosed neurofibromatosis type I: an uncommon cause of extrahepatic cholestasis. Z Gastroenterol. 2010;48:479–81. (PMID: 20352594)
- Subramanian M, Vikram A, Sankar S, Arunkumar et al. Sri Ramchandra Journal of Medicine, 2006 Sept 1.
- Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130:1466–78. (PMID: 17090188)
|