48uep6bbphidvals|579
48uep6bbph|2000F98CTab_Articles|Fulltext
Introduction
There are a number of case reports of acute pancreatitis associated with organophosphorus poisoning (1-5). A cause and effect relationship has been demonstrated in animal studies (6). However this association is perhaps still not widely recognized. Neither do commonly used surgical and medical textbooks describe acute pancreatitis as a presenting feature of organophosphorus poisoning nor is organophosphorus poisoning listed as one of the aetiological factors of acute pancreatitis and its complications. In adults the frequency of acute pancreatitis related to organophosphorus poisoning is 12.7% (7). Organophosphorus poisoning is not uncommon in our society; given the widespread availability and use of organophosphorus insecticides. Poisoning may be ccupational, accidental or suicidal. Tropical Gastroenterology 2013;34(1):44–47
Case Report
A male patient aged about 18 years was admitted in the surgical emergency of Nehru Hospital, B.R.D. Medical College, Gorakhpur with a two-day history of severe abdominal pain, abdominal distension, vomiting, palpitation and fever. Ten days prior to this admission, he was admitted in the medical emergency of the same hospital following ingestion of two teaspoons of organophosphate insecticide (Feridon) in a suicide attempt. He was managed conservatively and was discharged after improvement. There was no past history of trauma, alcoholism or pancreaticobiliary disease, nor significant medication history.
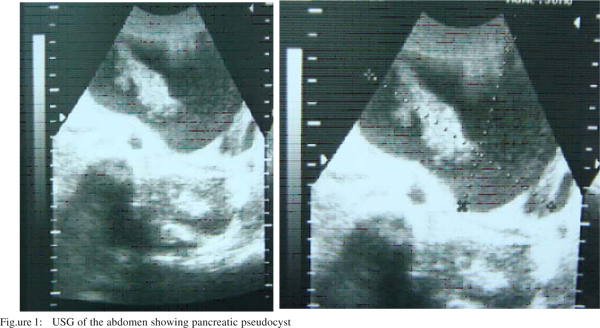
On examination, he was dehydrated, anemic, febrile, tachycardic, tachypneic, hypotensive, and had upper abdominal distension and rigidity all over the abdomen. Intravenous medications were started and investigations revealed hemoglobin 9 g/dL, total leucocytes count 16000/cumm, differential count- polymorphs 80% lymphocytes 10% eosinophils 6% and monocytes 4%, serum alkaline phosphatase 412 IU/L, serum amylase 800 IU/L, lactic dehydrogenase 1200 IU/L, serum lipase 560 IU/L, serum calcium 7 mg/dL, blood urea 54 mg/dL and serum creatinine 1.6 mg/dL. Other laboratory findings were near normal range. Electrocardiogram was normal excepting the tachycardia. Radiological investigations revealed obliterated left costophrenic angle in X-ray chest, features of peritonitis (ground-glass appearance) in X-ray erect abdomen and bulky pancreas with peripancreatic cyst of size 12 cm x 8 cm x12 cm with mild ascites on ultrasonography (USG).
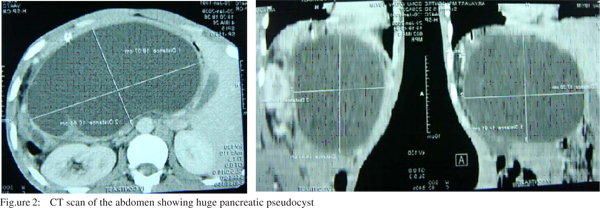
Patient was managed conservatively and became asymptomatic except upper abdominal distension but oral feeding was well tolerated. He was discharged from the hospital with advice to follow up in the OPD with serial hemoglobin and ultrasonography (Figure 1). At six-week follow up his other radiological findings became normal but the size of the cyst gradually progressed. He was readmitted and a dynamic computerized tomography (CT) scan was performed which showed diffusely bulky pancreas measuring 2.1 cm, 3.4 cm, and 2 cm in head, body and tail regions, respectively with huge pancreatic pseudocyst of size 18 cm x 10 cm x 15 cm in transverse x AP x cranio-caudal diameter with minimal ascites (Figure 2). After correcting anemia by blood transfusion and further preoperative assessment and treatment, he underwent exploratory laparotomy and internal drainage of the cyst in the form of cystogastrostomy. A postoperative ultrasonography was performed to ensure complete drainage of the cyst. His postoperative recovery was uneventful. The patient is under regular follow up for the last nine months and has no complaints till date.
Discussion
Organophosphorus compounds are commonly used worldwide as agricultural insecticides and in this region to eliminate household insects (mosquitos and cockroaches). Acute pancreatitis following organophosphorus poisoning generally shows a subclinical course (8). Organophosphate insecticides are potent inhibitors of enzyme acetylcholinesterase which leads to increased acetylcholine activity responsible for systemic symptomatology seen in organophosphorus poisoning such as hypersialorrhoea, pinpoint pupils, cramping abdominal pain and vomiting. They directly stimulate secretion of pancreatic juice and trypsin. The pancreatic duct and bile duct pressure increases because of over secretion of pancreatic juice and ampullary spasm, pancreatic duct spasm and edema. This hyper-stimulation has been hypothesized as the mechanism of acute pancreatitis following organophosphorus poisoning (9-11). In 1979, Dressel (12) described the first case report of pancreatitis following organophosphorus poisoning.
A pancreatic pseudocyst develops within a period of 1-4 weeks following the onset of acute pancreatitis as a result of pancreatic enzymes, debris, fluid, tissues and blood collection. The incidence of pancreatic pseudocyst development in acute pancreatitis is 15% (13).
Abdominal pain that may or may not radiate to the back is a common symptom in patients. Serum pancreatic enzymes and suitable imaging techniques are used more often in determining the patient’s clinical condition. The vast majority of pancreatic pseudocysts resolve spontaneously, however a cyst that does not resolve spontaneously (10% of cases) may lead to serious complications such as pain, pseudocyst rupture and abscess formation due to the expanding size of the lesion and compression over viscera (14). In our patient acute pancreatitis was suspected due to history of admission a week previous, in the medical emergency with abnormal pancreatic enzymes following organophosphorus poisoning. Pancreatic pseudocyst was diagnosed by ultrasonography. Possible etiological factors for acute pancreatitis (alcoholism, biliary diseases, medication and others) were excluded. Due to the history of ingestion of organophosphate insecticide, we diagnosed pancreatic pseudocyst as a complication of organophosphate induced pancreatitis. We initially managed the case conservatively with the help of serial ultrasonography. However, because of the progressive abdominal distension we did dynamic CT scan of the abdomen. Subsequently the patient underwent exploratory laparotomy and internal drainage (cystogastrostomy), which was followed by complete recovery.
In conclusion one should keep in mind that this complication can arise during the course of organophosphorus poisoning management and appropriate surgical treatment is necessary to reduce the morbidity and mortality.
References
1. Moore PG, James OF. Acute pancreatitis induced by organophosphate poisoning? Postgrad Med J. 981;57:660–2.
2. Dagli AJ, Moos JS, Shaik WA. Acute pancreatitis as a complication diazinon poisoning. J Assoc Physicians India. 1981;29:794–5.
3. Dagli AJ, Shaik WA. Pancreatic involvement in malathionanticholinesterase insecticide intoxication. A study of 75 cases. Br J Clin Pract. 1983;37:270–2.
4. Lee HS. Acute pancreatitis and organophosphate poisoning. A case report and review. Singapore Med J. 1989;30:599–601.
5. Lankisch PG, Muller CH, Neiderstadt H, Brand A. Painless acute pancreatitis subsequent to Anticholinesterase insecticide (Parathion ) intoxication. Am J Gastroenterol. 1990;85:872–5.
6. Dressel TD, Goodale RL, Zweber B, Borner JW. The effect of atropine and duct decompression on the evolution of Diazinoninduced acute canine pancreatitis. Ann Surg. 1982;195:424–34.
7. Sahin I, Onbasi K, Sahin H, Karakaya C, Ustun Y, Noyan T. The prevalence of pancreatitis in organophosphate poisoning. Hum Exp Toxicol. 2002;21:175–7.
8. Harputluoðlu MM, Kantarceken B, Karincaoglu M, Aladag M, Yildiz R, Ates M, et al.Acute pancreatitis: an obscure complication of organophosphate poisoning. Human Exp Toxicol. 2003;22:341–3.
9. Goodale RL, Manivel JC, Borner JW, Liu S, Judge J, Li C, et al. Organophosphate sensitizes the human pancreas to acinar cell injury. An ultrastructural study. Pancreas. 1993;8:171–5.
10. Brahmi N, Blel Y, Kouraichi N, Abidi N, Thabet H, Amamou M. Acute pancreatitis subsequent to voluntary methomyl and dichlorvos intoxication. Pancreas. 2005;31:424–7.
11. Kandalaft K, Liu S, Manivel C, Borner JW, Dressel TD, Sutherland DE, et al. Organophosphate increases the sensitivity of human exocrine pancreas to acetylcholine. Pancreas. 1991;6:398–403.
12. Dressel TD, Goodale RL, Amerson MA, Borner JW. Pancreatitis as a complication of Anticholinesterase Insecticide Intoxication. Ann Surg. 1979;189:199–204.
13. Isselbacher KJ, Braunwald E, Wilson JD. Harrison’s Principles of Internal Medicine Companion Handbook. 13th ed. Singapore: McGraw-Hill, Inc 1995;514–6.
14. Andrén-Sandberg A, Dervenis C. Pancreatic pseudocyst in the 21st century. Part II: natural history. JOP. 2004;5:64–70.