48uep6bbphidvals|395
48uep6bbph|2000F98CTab_Articles|Fulltext
Introduction
Enteric duplication cyst is an uncommon malformation of the gastrointestinal tract which is a diagnostic and therapeutic challenge. It can occur anywhere from mouth to anus, of which ileum is the commonest site. It is either asymptomatic or presents with vague symptoms mimicking other more common pathologies and is encountered unexpectedly intraoperatively. It is most commonly diagnosed when complications such as bleeding, intestinal obstruction or perforation occur. This is a case report of a patient with infected ileal duplication cyst who presented with acute intestinal obstruction and peritonitis.
Case Report
A 4 year old female child was brought to our emergency with history of fever for 1 week and bilious vomiting, abdominal distension and obstipation for 5 days. There was history of intermittent colicky abdominal pain. There was no history suggestive of worm infestation, viral infections or abdominal trauma. Patient was treated as typhoid fever with peritonitis for 3 days and later referred to us. On examination the child was dehydrated, febrile with tachycardia and hypotension. Abdominal examination showed grossly distended abdomen with visible intestinal peristalsis. There was abdominal guarding, rigidity and diffuse tenderness.. Bowel sounds were present. Per-rectal examination showed an empty rectum with anterior wall tenderness. Blood investigation showed elevated white cell count (24,000/ mm3) with predominance of polymorphs (78%). There was hypokalemia which was corrected before surgery. Blood widal for typhoid fever was negative and serum amylase was normal. Plain X- ray abdomen erect showed multiple air fluid level with absent colonic gas or pneumoperitoneum. Ultrasonogram of abdomen showed free fluid of about 50 ml in the pelvic cavity with grossly dilated bowel loops with no mass lesion. With the impression of small bowel obstruction with peritonitis of unknown etiology, the child was taken up for emergency laparotomy via right transverse supra-umbilical incision after adequate resuscitation.
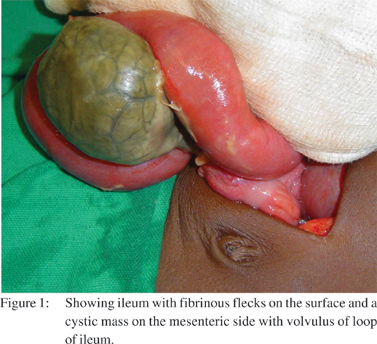
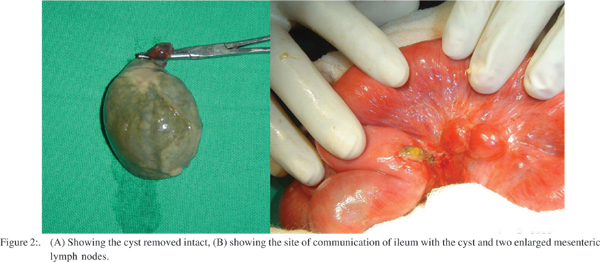
There was about 50 ml of pus in the pelvic cavity with fibrinous flecks in the bowel surface causing flimsy adhesions, which were released. A sample of pus was sent for Gram staining and culture. There was a 10 x 8 cm pear shaped tan–brown, leathery, tense cystic mass attached to the mesenteric side of the ileum (Figure 1). It was communicating with the lumen of the ileum through a narrow pedicle. It was located 30 cm proximal to the ileocecal junction. There were few blood vessels seen on the serosal surface of the cyst with no blood flow. There was a contained perforation in the base of the cyst close to the ileal wall sealed by fibrinous flecks. There was 270 degree volvulus of loop of ileum with the mass without any bands. The cyst was free from the mesenteric layers excluding a mesenteric cyst. There were no separate blood vessel or band(s) attached to the mass. The mass could be completely excised without difficulty after derotation of the volvulus (Figure 2A). The communicating edge of the ileum was closed with interrupted absorbable sutures after trimming the edges, which were sent for histopathology. There were two enlarged mesenteric lymph nodes in the vicinity of the cyst which were biopsied (Figure 2B). The lumen of the cyst contained feculent smelling turbid brown fluid and gummous material. Its interior surface was shaggy, irregular in thickness with no septae or mucosal lining recognizable on gross examination. A peritoneal lavage was done and abdomen closed with a peritoneal drain. The post operative period was uneventful and patient was discharged on 5th postoperative day. Gram staining and culture of pus was negative for any bacterial organism. Histopathologic examination of the cyst showed infarcted and necrotic mucosal lining, with plenty of acute inflammatory cells comprising of neutrophils, eosinophils and plasma cells, seen in all layers.
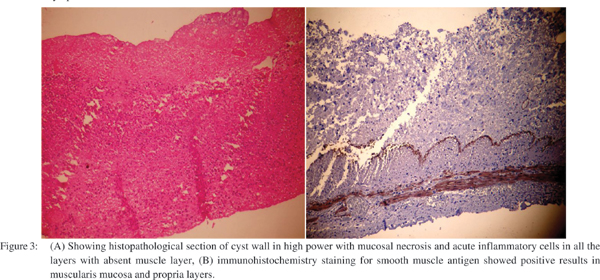
The submucosa showed congested blood vessels. No muscle layer could be made out in initial paraffin sections and on thinner multiple sections few atrophic, thin fibers with lost architecture were made out (Figure 3A). Thus the possibility of the cyst being a duplication cyst was remote. To exclude a duplication cyst, immunohistochemistry for smooth muscle antigen (SMA) was done which showed presence of smooth muscles in the muscularis mucosa and muscularis propria layer (Figure 3B). The presence of smooth muscle distinguished the lesion from a mesenteric cyst, meconium cyst and pseudocyst. There was no malignancy or dysplasia. The communication point with the ileum was lined by ulcerated intestinal mucosa with inflamed granulation tissue, a finding consistent with the presence of an inflammatory fistulous tract. There was no gastric mucosa or peptic ulceration in the communication site or in the cyst. There was reactive hyperplasia of mesenteric lymph nodes. At three months of follow up the patient was normal.
Discussion
Duplications of the alimentary tract are rare congenital anomalies that can present a diagnostic as well as therapeutic challenge. Since these lesions can be encountered unexpectedly intraoperatively, appropriate surgical management requires that the surgeon be familiar with the anatomy and clinical characteristics of these lesions.1 Congenital cystic masses in relation with mesentery includes mesenteric cyst, duplication cyst, lymph cyst, meconium cyst, etc. The characteristic features which help to differentiate duplication cyst are its location on the mesenteric side of bowel, the presence of smooth muscle in its wall lined by glandular mucosa that contains one or more cell types of the gastrointestinal tract.[2] Duplications may be mistakenly diagnosed as mesenteric or omental cysts or cystic lymphangiomas, but these lesions have endothelial rather than mucosal linings.[1] The first use of the term “duplications” has been attributed to Reginald Fitz, who described the lesions in 1884 and thought they were remnants of the omphalomesenteric duct.[3] Although its pathogenesis has not been established several theories have been proposed to explain the pathogenesis of duplications, including the persistence of embryonic diverticula, epithelial sequestration, the sparing of a bowel segment as a result of embryonic vascular insufficiency or trauma, and anomalous development of a duplicate lumen due to an error in recanalization after the solid phase of enteric mucosal development in the fetus.[3] It is possible, however, that duplications at different anatomical sites develop through different mechanisms.
Duplication cysts can occur anywhere in the gastrointestinal tract, from the tongue to the anus and most commonly the ileum.[2] They may be single or multiple, and spherical or tubular, varying widely in size. Spherical lesions usually do not communicate with the adjacent intestine are usually filled with clear secretions. In contrast, tubular duplications vary in length and can involve long segments of intestine and may have communication at proximal, distal or both ends and may become filled with bowel contents. The manifestations of intestinal duplications vary. Their size, location and complications determine the signs and symptoms.[1]
They may be asymptomatic or manifest with a palpable abdominal mass, abdominal pain, maelena, vomiting, and an acute abdominal presentation with intestinal obstruction, which may be complicated by a volvulus or intussusception.[4] They are difficult to diagnose if they have both a proximal and a distal opening and if there is no obstruction. Ectopic gastric mucosa is present in approximately 50 percent of duplications which can cause peptic injury leading to mucosal ulceration and hemorrhage into the adjacent bowel.[4] Occasionally, the entire cyst can becomes necrotic and lead to fistula formation with the adjacent intestine, peritoneum, or other nearby structures can also occur. Presence of only proximal communication with the bowel or formation of a fistula due to peptic ulceration may lead to dilatation, stagnation of the contents and infection of the cyst which may enlarge to such an extent that it mimics chylous ascites.[5] Secondary infection of the cyst leads to increase in pressure and ischemia and eventually perforation can occur as in our case.[5] With either infection or hemorrhage, the contents of the cyst change from clear fluid to turbid fluid or a semisolid material, with a consequent change in the features of the cyst’s appearance.
These changes in the character can even confuse the diagnosis per-operatively. In our case the clinical presentation mimicked a typhoid perforation of ileum which is a common scenario in the Indian subcontinent.[6] Per-operatively the appearance was suggestive of either a meconium cyst or an infected vitellointestinal duct cyst, a chylolymphatic mesenteric cyst or a fecal pseudocyst due ileal perforation. Meconium cyst was considered because of its appearance, close association with bowel and communication with the bowel, though there was no calcification of the wall.[7] Location on the mesenteric side, absence of any band connecting it with the umbilicus, made vittelline duct cyst unlikely. Chylolymphatic cyst was unlikely as mesenteric leaves were intact and the cyst could be separated without need for bowel resection.
Complicated duplication cyst presenting with infection or abscess is very rare and only few cases are reported. It has been reported that the inflammatory mass caused by duplication cyst has been mistaken for an inflammatory mesenteric mass, lymphoma, pelvic adnexal mass, pseudocyst, etc.[8,9,10,11] A case of infected ileal duplication cyst simulating acute appendicitis and peritonitis similar to our case has been described.[12,13] The organism found in cases of infected duplication cyst were Salmonella spp., Mycobacterium avium complex and Candida spp.; of course no microbiological predilection has been documented.[9,14,15] In our case there was no specific microorganism grown. The sonographic ‘double wall sign’ or ‘muscular wall sign’ is pathognomonic of duplication cyst and also the cornerstone in diagnosis. The double wall sign can be lost if there is infection and leads to erroneous diagnosis.[16]
The potential sonographic visualization of the split hypoechoic muscularis propria layer or identification of all five layers will increase the specificity in making the sonographic diagnosis of duplication cyst.[16] The infection of cyst and grossly dilated bowel due to ileal volvulus would have caused false negative impression of absent mass lesion in our case. Computerized tomogram would have helped but we have not done a CT, because the clinical manifestation warranted an emergency abdominal exploration. The presence of heterotopic gastric tissue is a primary cause of perforation that has been reported in as many as one third of patients with small intestine duplications. However, the detection of such ectopic tissue is time-consuming, and there is no readily available method of diagnosing tubular duplications. In our case there was no gastric mucosa but still there was a sealed perforation of the cyst
which would have occurred due to complete necrosis of the wall due to infection and ischemia.
References
1. Iyer CP, Mahour GH. Duplications of the alimentary tract in infants and children. J Pediatr Surg. 1995;30:1267–70.
2. Bower RJ, Sieber WK, Kiesewetter WB. Alimentary tract duplications in children. Ann Surg. 1978;188:669–74.
3. Holcomb GW, 3rd, Gheissari A, O’Neill JA, Jr., Shorter NA, Bishop HC. Surgical management of alimentary tract duplications. Ann Surg. 1989;209:167–74.
4. Tanabe ID, DiTomaso A, Pinkas H, Pencev D. Massive GI hemorrhage from an ileal duplication cyst in an adult. Am J Gastroenterol. 1995;90:504–5.
5. Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 8-2000. An 8 1/2 year-old girl with a painful abdominal mass. N Engl J Med. 2000;342:801–6.
6. Lalitha MK, John R. Unusual manifestations of salmonellosis – a surgical problem. Q J Med. 1994;87:301–9.
7. Herman TE, Siegel MJ. Meconium periorchitis. J Perinatol. 2004;24:53–5.
8. Lim GY, Im SA, Chung JH. Complicated duplication cysts on the ileum presenting with a mesenteric inflammatory mass. Pediatr Radiol. 2008;38:467–70.
9. Trojan J, Mousset S, Caspary W. F, Hoepffner N. An infected esophageal duplication cyst in a patient with non-Hodgkin’s lymphoma mimicking persistent disease. Dis Esophagus 2005;18:287–9
10. Caspi B, Schachter M, Lancet M. Infected duplication cyst of ileum masquerading as an adnexal abscess—ultrasonographic features.J Clin Ultrasound. 1989;17:431–3.
11. Hwang IK, Namkung S, Kim BS, Kim HC, Lee IS, Hwang WC. Perforated ileal duplication cyst with haemorrhagic pseudocyst formation. Pediatr Radiol. 2003;33:489–91.
12. Sakamoto K, Hasegawa S, Yamazaki Y, Makino T, Suda T, Imada T. Ileal duplication presenting as perforation: report of a case. Surg Today. 2000;30:445–7.
13. Islah MA, Hafizan T. Perforated ileal duplication cyst presenting with right iliac fossa pain mimicking perforated appendicitis. Med J Malaysia. 2008;63:63–4.
14. Jancelewicz T, Simko J, Lee H. Obstructing ileal duplication cyst infected with Salmonella in a 2-year-old boy: a case report and review of the literature. J Pediatr Surg. 2007;42:E19–21.
15. Kuwashima S, Chikatsu H, Kohno T, Fujioka M, Hagisawa S, Tsuboi T. Esophageal duplication cyst complicated by Mycobacterium avium complex infection. Pediatr Int. 2005;47:592–4.
16. Cheng G, Soboleski D, Daneman A, Poenaru D, Hurlbut D. Sonographic pitfalls in the diagnosis of enteric duplication cysts. AJR Am J Roentgenol. 2005;184:521–5.