|
|
|
|
 |
 |
| |
 |
|
|
Original Articles |
|
|
|
|
|
Keywords :
Neuroendocrine tumors, carcinoid syndrome. |
|
|
Amarapurkar DN1, Juneja MP1, Patel ND1, Amarapurkar AD2, Amarapurkar PD1
*Department of Gastroenterology & Hepatology, Bombay Hospital
Medical Research centre1
*Department of Pathology,
BYL Nair Ch Hospital2, Mumbai
Corresponding Author:
Dr. Deepak N. Amarapurkar
Email: amarapurkar@gmail.com
DOI:
http://dx.doi.org/
Abstract
Aim: Gastrointestinal tract is the commonest site for neuroendocrine tumors. Appendix, ileum and rectum were considered to be common sites for these tumors. However, there has been change in pattern of gastrointestinal neuroendocrine tumors over last few years. There is limited data available on epidemiology and patterns of these tumors in . Methods: Analysis of 74 patients with gastrointestinal and pancreatic neuroendocrine tumors over a period of 7 years at a single center in
Mumbai, was done. Clinical details, surgical outcome with follow up and treatment were reviewed. All these patients were analyzed with special emphasis on the site of the tumor.
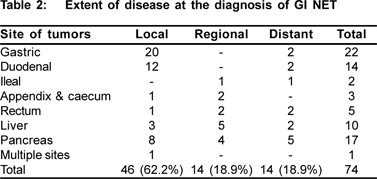
Results: The results showed a male preponderance (ratio of 2.5:1) with a mean age of 53.01 ± 15.13 years. Of the 74 tumors, the commonest site was found to be stomach 22 (30.2%), followed by pancreas 17 (23.3%) and duodenum 14 (18.9%). Only 3 (4.1%) patients presented with carcinoid syndrome. The disease was localized in 46 (62.2%), regional spread was seen in 14 (18.9%) and distant spread in 14 (18.9%). Majority of gastric and duodenal tumors had localized disease while pancreatic NETs led to most of the cases with distant disease.
Conclusion: This analysis showed that gastrointestinal and pancreatic neuroendocrine tumors are not rare. Pattern of these tumors has definitely changed over last few years. Stomach was found to be commonest site for gastrointestinal neuroendocrine tumors followed by pancreas and duodenum.
|
48uep6bbphidcol4|ID 48uep6bbphidvals|306 48uep6bbph|2000F98CTab_Articles|Fulltext The estimated prevalence of neuroendocrine tumors (NET) is 1 to 2 cases per 100,000 people, of which gastrointestinal (GI) tract is the most common site.[1] The incidence of GI –NET is around 67.5% amongst all NET.[2] Majority of these tumors have an indolent course. Some are diagnosed incidentally while few have disseminated disease and may present as metastatic disease. The criteria for establishing the degree of malignancy in carcinoids tumor remains unclear. Histological analysis often fails to distinguish the aggressive and metastatic potential of the tumor. GI NETs show varying biology and present sometimes with distinct clinical syndromes such as flushing, diarrhoea, hypoglycemia and gastric ulcers. Amongst the GI – NET’s, appendix and ileum followed by rectum were considered to be the most favourite sites.[3,4] However, the trend appears to be changing over last few years.[5,6,7,8] There is limited data available on epidemiology of GI – NET in India. This case series examined patterns, incidence and outcomes of GI NET from tertiary referral centre in India.
Materials and Methods
This was a retrospective as well as prospectively maintained data analysis of GI and pancreatic neuroendocrine tumors, from a single gastroenterology unit of tertiary referral hospital over a period of 7 years. Records of patients diagnosed as GI NET on the basis of histopathology, maintained by one of the authors over a period of 7 years were reviewed. History with clinical details were obtained from records maintained by the same person. Data regarding routine laboratory investigations, abdominal ultrasound, CT Scan, MRI, upper or lower GI endoscopy and surgical records, medical treatment and follow up data were analyzed. Diagnosis of GI NET was based on histopathology with immune histochemical staining. In prospectively collected data, after the diagnosis of NET on endoscopy with biopsy, the patients were investigated for serum chromogranin, serum gastrin and radiological tests such as USG or CT scan. Parietal cell and intrinsic factor antibodies were done in patients with gastric NETs. 24 hrs urine 5HIAA was performed in clinically suspected cases for carcinoid syndrome. Patients were treated accordingly to site of tumor, spread of tumor, symptoms and patient’s preference.
Results
Over 7 years (2000 to 2007), a total of 74 cases were diagnosed as GI and pancreatic NET. Table 1 shows the demographic and presenting symptoms of these patients. The most commonly involved organs were stomach in 22 (30.2%) patients, followed by pancreas in 17 (23.3%) and duodenum in 14 (18.9 %). The demographical profile showed mean age of 53.01 ± 15.13 years (range:16-82). The youngest patient was 16 years male with pancreatic NET and the oldest patient was 82 years male with rectal NET. Majority of the patients were between 41 to 60 years of age. There were 52 males and 22 females with male to female ratio of 2.5: 1. The clinical presentation of patients varied in these patients (Table 1). Abdominal pain was the commonest presenting symptom in 30 (40.5 %) patients. Miscellaneous symptoms seen in 17 (23.0%) patients included itching, tremors, tingling numbness, anemia, hypoglycemia, skin lesions and restlessness. There were 3 asymptomatic patients who were cases of gastric NETs diagnosed incidentally and had gastric type 1 NET. Seven (9.5%) patients had Zollinger Ellison (ZE) syndrome of which 4 (5.4%) had multiple endocrine neoplasm (MEN) I. There was one patient (1.3%) who had MEN II. Only three (4.1%) patients presented with carcinoid syndrome.
From 22 gastric NETs, 16 (72.7%) were type I (associated with chronic gastritis and gastric atrophy), 4 (18.2%) were type II, associated with gastrinomas with or without MEN1. Two patients had Type III gastric carcinoids (sporadic tumors). Of 16 type 1 gastric NETs, 15 (93.8%) were males and 1 (6.2%) female. In type II both males and females were equal in proportion while both the cases of type III were males. On endoscopy all type I gastic NETs had multiple polyps less than 2 cms. All of these patients had gastric histology negative for H. Pylori infection, had B12 deficiency, elevated serum gastrin levels and parietal cell antibodies and intrinsic factor antibodies positive. Even though antrectomy or polypectomy is considered as treatment for type 1 gastric NET, conservative follow up is one of the choice of treatment for type 1 gastric carcinoids.[9,10,11] All these patients were treated with parenteral B12 supplements and followed up regularly. Follow up period ranged from 1 to 6 years. None of these patients developed malignancy in follow up period. Of the 4 patients with Type II Gastric NET, 3 patients underwent surgery for primary pancreatic tumor while one patient had extensive metastatic disease. Of the patients who underwent Whipple’s surgery for pancreatic tumor, one developed recurrence of tumor after 2 years of surgery and required second surgery. All the patients were symptom free and alive at the end of follow up. One patient with extensive metastatic disease was treated with long acting Octreotide (Sandostatin LAR). This patient succumbed 6 months after the diagnosis. Of the two sporadic gastric carcinoid, one tumor was resected with endoscopic mucosal resection. The second patient underwent surgical wedge resection; both patients are doing well (currently 9 months) after resection. Of the total 17 pancreatic NETs, 12 were functional and 5 were non functional tumors. Functional tumor types were gastrinoma in 7, glucaganoma in 1 VIPOMA 1, insulinoma in 3 patients. Four patients were associated with MEN I (pituitary tumor, parathyroid adenoma and pancreatic tumors).
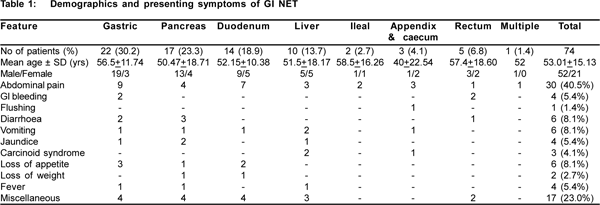
Of the 14 duodenal NETs, 3 (21.4%) were associated with a gastric NET type 1 (associated with chronic gastritis and gastric atrophy). 1 (7.1%) patient had gastrinoma with MEN1. Follow up ranged from 1 to 22 years and most of the patients have stable disease. The patient with gastrinoma with MEN1 has succumbed to the disease. Ten patients had primary liver carcinoids. Male and female distribution was equal with average age of presentation 51.5+18.17 years. Two out of 10 patients presented with carcinoid syndrome while 3 patients had abdominal pain. One had jaundice, 2 had vomiting and 3 other had other symptoms and detected on ultra sonography. Both patients with carcinoid syndrome had solitary tumor and underwent surgical resection and are without recurrence in follow up period for 3 to 4 years. Three patients had multiple (more than 50 lesions) distributed all over the liver treated with embolization without much benefit. All the three patients succumbed in one year. Remaining 5 patients had multiple lesions (2 to 6 lesions) treated with embolization and are doing well in follow up period of 1 to 6 years.
The disease was localized in 46 (62.2%) patients, regional spread was seen in 14 (18.9%) and distant spread in 14 (18.9%) patients. Majority of the gastric and duodenal NETs had localized disease (Table 2). Regional spread was common in liver and pancreas NETs. Pancreatic NETs led to most of the cases with distant disease.
Discussion Gastrointestinal and pancreatic NETs are rare malignant tumors. Due to improved diagnostic and therapeutic modalities, they have gained attention over last few years. From all over the world, there is limited epidemiological data available for GI and pancreatic NETs. From India there are very few reports available on epidemiology of these tumors.[12,13] Hence, this study was planned to determine the pattern of NET of GI tract and pancreas.
Amongst 74 GI and pancreatic NETs, majority of the patients were between 41 to 60 years group with mean age of 53 years. Tumors were more common in males than females (M:F = 2.5:1). Various reports in the literature have showed mean age for GI NETs to be around 55 years,[14,15].with equal prevalence in both males and female[16] Sometimes it has been shown to even be slightly more common in females.[17] However, in present series, males outnumber females. In a series of 3382 carcinoids of all types, Newton et al[18] reported an overall age adjusted incidence of 0.71 for men and 0.87 for women per 100,000 population per year. In Denmark, incidence for both men and women was 1.1 per 100,000 per year.[19]
Carcinoid syndrome is frequently discussed in relation to carcinoid tumors. However, the complex of flushing, diarrhoea, abdominal pain, and occasional asthma or right-sided valvular problems is actually uncommon: less than 10% (1.7%-8.4%) of carcinoids exhibit some of these symptoms.[20,21] In this study, there were only three (4.1%) patients presenting with carcinoid syndrome.
In contrast to other studies[7,17] in which 40% to 60% of tumors are found incidentally, carcinoid tumors detected incidentally represented only 4.1% of the tumors in our analysis. This most likely can be attributed to the tertiary nature of our medical center, where patients have often been referred for definitive diagnosis or treatment.
Over last few years appendix, ileum and rectum were considered to be the common sites for NET of GI tract.[3,4] In contrast in our study, stomach was the commonest site followed by pancreas and duodenum. This is very similar to recent epidemiological data suggesting that the incidence of gastric carcinoids have risen markedly over the last three decades.[5,6,16] The percentage of gastric carcinoid amongst all other NETs has increased from 2.25% in Godwin’s series[3] to 5.9 % in the latest SEER data base and remained at 8.7 % of the entire set of 13,715 carcinoid tumors.[2] Hodgson et al[16] in 2005 have shown statistically significant eight or nine fold increase in the incidence of gastric carcinoids from two large databases. Modlin et al[22] have shown significant increase in incidence of gastric carcinoids from 2.4 to 8.7 %. A study from India analyzing cases of gastric carcinoid has also shown rising incidence of gastric NETs as compared to the past.[12] This increase can be attributed to widespread use of proton pump inhibitors or increased endoscopic surveillance with expertise in reporting gastric biopsies. The association between higher serum gastrin levels, atrophic body gastritis with low acid output and gastric endocrine cell hyperplasia with subsequent development gastric NETs have been well documented. More detailed investigations such as endoscopy with biopsy in this subgroup of atrophic body gastritis, are useful in detecting gastric NET at an early stage.
Modlin et al[23] in 2007 have analysed largest number of small bowel NETs with incidence being 21% for small bowel NETs and 49.6% for ileal NET. They have analysed the pattern, incidence, prognosis and outcome of small intestinal NET over a 30 years period. We have come across only 2 (2.7 %) of small intestinal NET. While reviewing these cases they have shown that there is a significant increase in the incidence of small intestinal NET, even though with new diagnostic modalities the survival rate has remained unchanged over 30 years. The clinical relevance of small intestinal NET is that their symptomatology is generally not apparent until hepatic metastasis has occurred; hence, they are rarely diagnosed at an early stage. They have metastatic disease at the time of presentation.
Although appendiceal carcinoids have long been recognized as the most frequently occurring carcinoid tumors, their relative frequency has reduced over period of time. In this study, there were only 3 cases of appendiceal carcinoid. Sandor and Modlin[24] in 1998 have studied the epidemiology of 1570 appendiceal carcinoids which comprised 18.9 % of all carcinoids tumors. They have reported significant female predominance for appendiceal NET. As compared to other carcinoids, 5 year survival rate for appendiceal carcinoids was 85.9 % which was the highest among all types of carcinoid tumors.
Rectum is considered to be 3rd most common site for GI NET. The incidence ranges between 5 to 50% in various studies. We had only 5 (6.8%) cases of rectal NET. Soga[25] reviewed large number of rectal NETs and have found a male preponderance. Rectal NET show predominant submucosal invasion with high incidence of metastasis. They are infrequently associated with carcinoid syndrome. Mortality is relatively high, within 5 years after removal of the lesion.
There is limited data on pancreatic carcinoids, incidence being 0.73 % of all type of carcinoids within pan SEER data set.[26] Pancreatic carcinoids are known to have malignant behaviour of which 72 – 81% have non localized disease.[27] Overall survival rate is 37.5 %. In our study, 17 pancreatic NET were diagnosed with high index of suspicion. The diagnosis was done with the help of serological markers to determine their functional ability. Three of them were associated with MEN1 and 1 with MEN2 syndrome. Soga[27] in 2005 analysed largest (156) number of pancreatic NET, and have found that pancreatic NET exhibit high metastatic rate and extremely low 5 years survival rate].
Extra hepatic biliary system is another site for NET. Liver is the commonest organ for metastasis of carcinoid tumors. However, primary liver NET are very rare.[28,29] In this study, there were 10 (13.7%) cases of primary carcinoid tumors of the liver in which there was no primary found in any other organ after rigorous search.
Conclusion
In conclusion, a definite change in pattern is evident in these tumors over the last few years. Gastric (type I) and pancreatic NETs were found to be commonest as compared to other GI NETs.
References
1. Sippel RS, Chen H. Carcinoid tumors. Surg Oncol Clin N Am. 2006;15:463–78.
2. Modlin IM, Ley KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97:934–59.
3. Godwin JD 2nd. Carcinoid tumors. An analysis of 2837 cases. Cancer. 1975;36:560–9.
4. Saha S, Hoda S, Godfrey R, Sutherland C. Raybon K. Carcinoid tumors of the gastrointestinal tract: a 44 year experience. South Med J. 1989;82:1501–5.
5. Delle Fave G, Capurso G, Milione M, Panzuto F. Endocrine tumors of the stomach. Best Pract Res Clin Gastroenterol. 2005;19:659–73.
6. Burkitt MD, Pritchard DM. Review article: Pathogenesis and management of gastric carcinoid tumours. Aliment Pharmacol Ther. 2006;24:1305–20.
7. Zeitels J, Naunheim K, Kaplan EL, Straus F 2nd. Carcinoid tumors. a 37-year experience. Arch Surg. 1982;117:732–7.
8. Oberg K. Neuroendocrine gastrointestinal tumours. Ann Oncol. 1996;7:453–63.
9. Granberg D, Wilander E, Stridsberg M, Granerus G, Skogseid B, Oberg K. Clinical symptoms, hormone profiles, treatment, and prognosis in patients with gastric carcinoids. Gut. 1998;43:223–8.
10. Solcia E, Capella C, Sessa F, Rindi G, Cornaggia M, Riva C, Villani L. Gastric carcinoids and related endocrine growths. Digestion. 1986;35(Suppl 1):3–22.
11. Ramage JK, Davies AH, Ardill J, Bax N, Caplin M, Grossman A, et al. UKNETwork for Neuroendocrine Tumours: Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours. Gut. 2005, 54(Suppl 4):iv1–iv16.
12. Hegde V, Mohandas KM, Ramadwar M, Shukla P, Mehta S. Gastric carcinoids – a changing trend. Indian J Gastroenterol. 2003;22:209–11.
13. Radhakrishnan S, Subramoniam S. Colerectal carcinoids in South India. Trop Geogr Med. 1979;31:63–7.
14. Bhaduri AS, Prayaga AS, Patel DD, Balar DB. Carcinoid tumours: a collective review of the last twelve years. Indian J Pathol Microbiol. 1986;29:414–21.
15. Van Gompel JJ, Sippel RS, Warner TF, Chen H. Gastrointestinal carcinoid tumors: factors that predict outcome. World J Surg. 2004;28:387–92.
16. Hodgson N, Koniaris LG, Livingstone AS, Franceschi D. Gastric carcinoids: a temporal increase with proton pump introduction. Surg Endosc. 2005:19:1610–2.
17. Shebani KO, Souba WW, Finkelstein DM, Stark PC, Elgadi KM, Tanabe KK, et al. Prognosis and survival in patients with gastrointestinal tract carcinoid tumors. Ann Surg. 1999;229:815–21; discussion 822–3.
18. Newton JN, Swerdlow AJ, dos Santos Silva IM, Vessey MP, Grahame-Smith DG, Primatesta P, et al . The epidemiology of carcinoid tumours in England and Scotland. Br J Cancer. 1994;70:939–42.
19. Westergaard T, Frisch M, Melbye M. Carcinoid tumors in Denmark 1978-1989 and the risk of subsequent cancers. A populationbased study. Cancer. 1995;76:106–9.
20. Ito T, Tanaka M, Sasano H, Osamura YR, Sasaki I, Kimura W, et al Preliminary results of a Japanese nationwide survey of neuroendocrine gastrointestinal tumors. J Gastroenterol. 2007;42:497–500.
21. Soga J, Yakuwa Y, Osaka M. Carcinoid syndrome: a statistical evaluation of 748 reported cases. J Exp Clin Cancer Res. 1999;18:133–41.
22. Modlin IM, Lye KD, Kidd M. A 50-year analysis of 562 gastric carcinoids: small tumor or larger problem? Am J Gastroenterol. 2004;99:23–32.
23. Modlin IM, Champaneria MC, Chan KC, Kidd M. A three-decade analysis of 3,911 small intestinal neuroendocrine tumors: the rapid pace of no progress. Am J Gastroenterol. 2007;102:1464–73.
24. Sandor A, Modlin IM. A retrospective analysis of 1570 appendiceal carcinoids. Am J Gastroenterol. 1998;93:422–8.
25. Soga J. Carcinoids of the rectum: an evaluation of 1271 reported cases. Surg Today. 1997;27:112–9.
26. Percy C, Young JL Jr, Muir C, Ries L, Hankey BF, Sobin LH, et al. Cancer. Introduction. Cancer. 1995;75:140–6.
27. Soga J. Carcinoids of the pancreas: an analysis of 156 cases. Cancer. 2005;104:1180–7.
28. Fenwick SW, Wyatt JI, Toogood GJ, Lodge JP. Hepatic resection and transplantation for primary carcinoid tumors of the liver. Ann Surg. 2004;239:210–9.
29. Amarapurkar AD, Davies A, Ramage JK, Stangou AJ, Wight DG Portmann BC. Proliferation of antigen MIB-1 in metastatic carcinoid tumours removed at liver transplantation: relevance to prognosis. Eur J Gastroenterol Hepatol. 2003;15:139–43.
|
|
|
 |
|
|